No products in the cart.
Sale
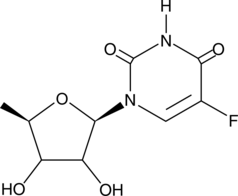
Doxifluridine | CAS 3094-09-5 | Nucleoside Analog for Cancer Research
Original price was: $3.00.$2.00Current price is: $2.00.
Doxifluridine is an orally administered nucleoside analog widely used in preclinical and laboratory research for its potential anti-proliferative and DNA synthesis-inhibiting effects. It serves as a prodrug of 5-fluorouracil (5-FU), undergoing metabolic conversion to release active 5-FU within cells, thereby interfering with nucleotide metabolism and DNA replication. Researchers employ doxifluridine to study cellular cytotoxicity, cancer cell response mechanisms, and combination strategies with other chemotherapeutic agents in vitro and in controlled laboratory models.
Description
Product Description
Doxifluridine is a fluorinated pyrimidine nucleoside analog developed for laboratory research to explore anticancer mechanisms, DNA replication inhibition, and nucleotide metabolism. As a prodrug of 5-fluorouracil, doxifluridine undergoes intracellular enzymatic conversion to release 5-FU, which is subsequently phosphorylated to its active nucleotide forms. These metabolites incorporate into RNA and DNA, disrupt nucleic acid synthesis, and inhibit thymidylate synthase, leading to S-phase cell cycle arrest and apoptotic pathways activation.
Researchers utilize doxifluridine to investigate the cellular response to nucleotide pool imbalance, DNA damage, and replication stress in various tumor cell lines. Its fluorinated structure enhances lipophilicity and oral bioavailability in vivo, but in laboratory research, it provides a controlled model for studying 5-FU-related cytotoxicity without direct administration of 5-FU. This makes it a valuable tool for mechanistic studies on drug metabolism, enzyme activation, and tumor cell sensitivity.
Experimental studies demonstrate that doxifluridine can modulate gene expression related to DNA repair, apoptosis, and nucleotide metabolism. Its activity is observed in both rapidly proliferating cancer cells and in models simulating chemotherapeutic resistance. By providing a controlled release of 5-FU intracellularly, doxifluridine offers insights into prodrug activation pathways and intracellular enzymatic kinetics, crucial for understanding fluoropyrimidine pharmacology at the cellular level.
Doxifluridine is widely employed in combination studies with DNA repair inhibitors, targeted small molecules, or other nucleoside analogs. This enables researchers to explore potential synergistic cytotoxicity, resistance mechanisms, and pharmacodynamic interactions. Its reproducibility in controlled laboratory assays makes it suitable for high-throughput screening and preclinical mechanistic research.
The compound’s chemical stability, ease of handling, and high purity allow for precise experimental design. Proper storage under low-temperature conditions and protection from light ensures retention of its biochemical integrity, supporting consistent and reliable laboratory results.
Product Specifications
| Item | Details |
|---|---|
| Product Name | Doxifluridine |
| CAS Number | 3094-09-5 |
| Synonyms | 5′-Deoxy-5-fluorouridine; 5′-DFUR |
| Molecular Formula | C9H11FN2O4 |
| Molecular Weight | 228.20 g/mol |
| Purity | ≥99% |
| Appearance | White to off-white crystalline powder |
| Solubility | Soluble in DMSO, water, and polar solvents |
| Storage Temperature | 2–8 °C |
| Category | Nucleoside analog |
| Applications | Cancer research, DNA synthesis inhibition, nucleotide metabolism, apoptosis studies |
| Formulation | Suitable for in vitro assays and preclinical laboratory research |
| Stability | Stable under recommended storage conditions |
| Shelf Life | 24 months |
| Supplier Type | Research chemical supplier |
| Intended Use | For laboratory research use only |
Mechanism of Action
Doxifluridine functions as a prodrug of 5-fluorouracil (5-FU) and primarily exerts its activity through nucleotide metabolism interference:
Intracellular Conversion and Metabolism
Doxifluridine is metabolized by intracellular enzymes, including thymidine phosphorylase, to release 5-FU. The released 5-FU is phosphorylated to active metabolites: 5-fluorodeoxyuridine monophosphate (FdUMP), 5-fluorouridine triphosphate (FUTP), and 5-fluorodeoxyuridine triphosphate (FdUTP). These active metabolites incorporate into RNA and DNA, disrupting nucleic acid function.Thymidylate Synthase Inhibition
FdUMP forms a stable ternary complex with thymidylate synthase (TS) and 5,10-methylenetetrahydrofolate, leading to irreversible inhibition of dTMP synthesis. This depletion of thymidine triphosphate pools halts DNA replication, particularly affecting rapidly proliferating cancer cells.RNA and DNA Incorporation
FUTP incorporates into RNA, causing defective RNA processing and protein synthesis. FdUTP incorporation into DNA induces strand breaks and triggers DNA repair pathways. This dual incorporation enhances cytotoxic effects and contributes to S-phase arrest.Apoptosis Induction
DNA damage and replication stress activate both p53-dependent and independent apoptotic pathways. Caspase activation, mitochondrial membrane depolarization, and chromatin condensation occur, leading to programmed cell death in susceptible tumor cell lines.Synergistic Mechanisms
In research, doxifluridine is often combined with other chemotherapeutics, DNA repair inhibitors, or signal transduction modulators to study potential synergistic cytotoxicity, drug resistance, and enhanced apoptosis induction.Research Implications
Doxifluridine provides a model for studying prodrug activation, enzyme kinetics, nucleotide pool imbalance, and tumor cell sensitivity to fluoropyrimidine-based treatments, facilitating mechanistic investigations and preclinical screening.
Side Effects
In laboratory research, doxifluridine demonstrates concentration-dependent cytotoxicity. Key considerations include:
Cellular Toxicity: Dose-dependent apoptosis and S-phase arrest in rapidly proliferating tumor cell lines.
DNA Damage: Incorporation into DNA can induce strand breaks, which is leveraged in mechanistic studies.
RNA Disruption: Interference with RNA processing and protein synthesis may affect normal cell cultures at high concentrations.
Safety Handling: Researchers must use proper PPE, avoid inhalation or skin contact, and adhere to institutional chemical safety protocols.
Storage Considerations: Protect from light, moisture, and high temperatures to maintain biochemical integrity.
Usage Limitation: For laboratory research only; not intended for therapeutic use.
Keywords
Doxifluridine, 5′-Deoxy-5-fluorouridine, nucleoside analog, DNA synthesis inhibitor, cancer research, apoptosis, laboratory research chemical, high-purity nucleoside, preclinical cytotoxicity, tumor cell studies, research compound supplier.
Shipping Guarantee
All shipments are handled using validated cold-chain logistics to preserve compound integrity. Each package is sealed in moisture-proof containers with secondary protective wrapping and continuous temperature monitoring. Products are shipped via express international couriers with full tracking and insurance coverage.
Trade Assurance
We ensure product authenticity, verified ≥99% purity, and compliance with analytical standards (HPLC, MS, NMR). Each batch is supplied with a Certificate of Analysis (CoA). Our trade assurance policy guarantees replacement or refund for any deviation from listed specifications.
Payment Support
We provide flexible and secure global payment options to support international research transactions. Accepted payment methods include PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). All transactions are protected by industry-standard encryption and verified payment gateways to ensure confidentiality and fund security.
Disclaimer
All products listed are intended for laboratory research use only and not for human or veterinary use. They are not drugs, medical devices, or diagnostics and should not be administered to humans or animals. Researchers must handle all materials in accordance with institutional biosafety and chemical safety guidelines. The information provided is for scientific reference only and does not imply therapeutic efficacy, safety, or regulatory approval.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
Q1: What is Doxifluridine used for in research?
A1: Doxifluridine is used as a nucleoside analog to study DNA synthesis inhibition, apoptosis, and chemotherapeutic mechanisms in preclinical cancer models.
Q2: What is the purity of Doxifluridine?
A2: Our Doxifluridine is supplied at high purity ≥99%, verified by HPLC, MS, and NMR.
Q3: How should Doxifluridine be stored?
A3: Store at 2–8°C short-term, or −20°C long-term, protected from light and moisture.
Q4: Is Doxifluridine water-soluble?
A4: Yes, it is soluble in water, DMSO, and other polar solvents, suitable for in vitro experiments.
Q5: Can it be used in vivo?
A5: Doxifluridine is for laboratory research only, with in vivo studies limited to controlled preclinical research.
Q6: How does Doxifluridine inhibit DNA synthesis?
A6: Through intracellular conversion to 5-FU, forming FdUMP, which inhibits thymidylate synthase, depleting thymidine pools and halting DNA replication.
Q7: Does it induce apoptosis?
A7: Yes, DNA damage and replication stress trigger both p53-dependent and independent apoptotic pathways in sensitive tumor cells.
Q8: Can it be combined with other drugs?
A8: Yes, it is frequently used with DNA repair inhibitors, nucleoside analogs, or chemotherapeutic agents for synergy and resistance studies.
Q9: Are analytical verifications provided?
A9: Yes, each batch includes a Certificate of Analysis (CoA) confirming ≥99% purity and analytical compliance.
Q10: Is it suitable for nucleotide metabolism studies?
A10: Absolutely. It is commonly used to study thymidylate synthase inhibition, DNA repair pathways, and nucleotide pool imbalance.
Q11: Can I purchase in bulk?
A11: Yes, bulk and OEM supply options are available for research laboratories.
Q12: What safety measures are needed?
A12: Use standard lab safety procedures: PPE, avoid inhalation or skin contact, and follow institutional chemical safety protocols.
Q13: What experimental models is it compatible with?
A13: Suitable for in vitro cell culture models, including human and rodent tumor lines.
Q14: Is it used for drug resistance research?
A14: Yes, it helps investigate mechanisms of resistance to fluoropyrimidine-based chemotherapy.
Q15: Do you offer OEM supply for research labs?
A15: Yes, we provide custom OEM production, bulk supply, and private labeling for research-grade Doxifluridine.











Reviews
There are no reviews yet.