No products in the cart.
Sale
Altretamine | CAS 645-05-6 | Alkylating Antineoplastic Agent for Laboratory Research
Original price was: $3.00.$2.00Current price is: $2.00.
Altretamine is a synthetic alkylating agent used in laboratory and preclinical research to study DNA crosslinking, cytotoxicity, and antineoplastic mechanisms. It is primarily applied in experimental models of ovarian cancer and other solid tumors to investigate chemotherapeutic responses and resistance pathways.
Description
Contents
hide
Product Description
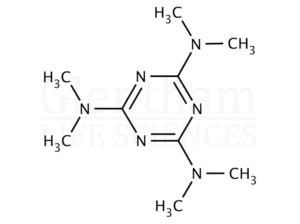
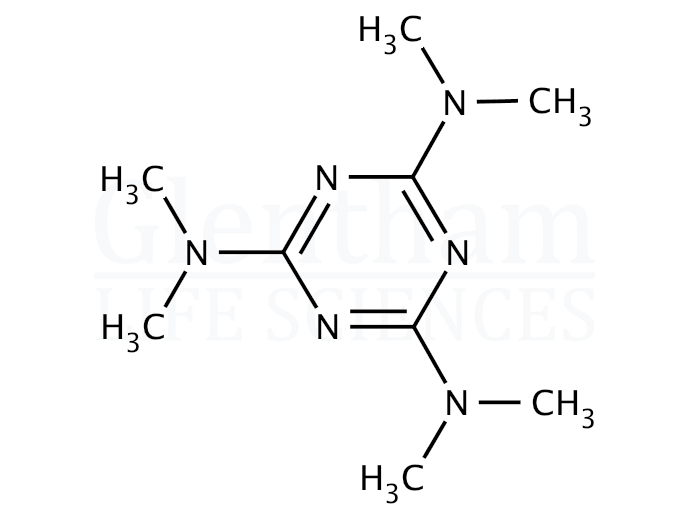
Altretamine, also known as hexamethylmelamine, is a lipophilic triazine derivative that functions as an alkylating cytotoxic agent in experimental oncology research. Its mechanism involves the formation of DNA crosslinks, resulting in replication inhibition, transcriptional interference, and subsequent tumor cell apoptosis.
This compound is widely used in laboratory studies to explore chemotherapeutic efficacy, DNA damage response pathways, and drug resistance mechanisms. Researchers employ Altretamine to investigate the sensitivity of various cancer cell lines, including ovarian carcinoma and melanoma models, to alkylating cytotoxic stress.
In preclinical models, Altretamine administration leads to the induction of DNA adducts and interstrand crosslinks, which disrupt the normal DNA replication process. Its alkylation preferentially occurs at guanine and adenine bases, contributing to the cytotoxicity observed in proliferating tumor cells. The study of Altretamine allows researchers to dissect pathways of DNA repair, apoptotic signaling, and cell cycle checkpoint activation.
Due to its lipophilic nature, Altretamine readily penetrates cellular membranes, ensuring intracellular access to nuclear DNA. In laboratory conditions, it has been demonstrated to induce cell cycle arrest primarily at the G2/M phase, providing a useful tool to study cell division regulation and chemotherapeutic intervention strategies.
Altretamine also serves as a model compound for examining combinatorial chemotherapy regimens, often studied in conjunction with platinum-based agents, topoisomerase inhibitors, or microtubule-targeting drugs to evaluate synergistic cytotoxicity in experimental settings.
Product Specifications
| Item | Details |
|---|---|
| Product Name | Altretamine |
| CAS Number | 645-05-6 |
| Synonyms | Hexamethylmelamine, HMM |
| Molecular Formula | C_12H_27N_3 |
| Molecular Weight | 237.37 g/mol |
| Purity | ≥99% |
| Appearance | White to off-white crystalline powder |
| Solubility | Soluble in DMSO and ethanol |
| Storage Temperature | 2–8 °C, protected from light |
| Category | Alkylating antineoplastic agent |
| Applications | DNA crosslinking studies, cytotoxicity assays, chemoresistance research, preclinical cancer modeling |
| Formulation | Lyophilized powder or solution for laboratory experiments |
| Stability | Stable under recommended storage conditions |
| Shelf Life | 24 months |
| Supplier Type | Laboratory research chemical supplier |
| Intended Use | For laboratory and preclinical research only |
Mechanism of Action
Altretamine acts as an alkylating agent that induces DNA damage in proliferating tumor cells. Its primary mechanism of action involves covalent binding to nucleophilic sites on DNA, leading to interstrand and intrastrand crosslinks.
1. DNA Alkylation and Crosslink Formation
Altretamine forms electrophilic intermediates that alkylate guanine and adenine residues in DNA. The resultant crosslinks impede DNA replication and transcription, leading to replication fork stalling and cell cycle arrest. This cytotoxic mechanism is central to its antineoplastic activity in experimental models.
2. Induction of Apoptosis
DNA crosslinking triggers DNA damage response pathways, including activation of ATM/ATR kinases and p53-mediated apoptotic signaling. The accumulation of DNA lesions leads to programmed cell death in sensitive tumor cell populations.
3. Cell Cycle Disruption
Altretamine treatment commonly results in G2/M cell cycle arrest. This arrest allows the accumulation of DNA damage and provides a platform for studying checkpoint control and repair mechanisms in laboratory models.
4. Synergistic Experimental Applications
In preclinical research, Altretamine is often studied in combination with platinum-based compounds or other chemotherapeutics to evaluate synergistic effects, potential enhancement of cytotoxicity, and overcoming chemoresistance mechanisms.
5. Model Compound for Mechanistic Studies
Due to its well-characterized DNA crosslinking activity, Altretamine serves as a valuable tool for studying DNA repair pathways, including nucleotide excision repair, homologous recombination, and mismatch repair, in both in vitro and in vivo research models.
Side Effects
Altretamine is strictly for laboratory and preclinical research and not intended for human use. Potential experimental considerations include:
Cytotoxicity to cultured cells requiring precise dose titration;
Potential instability in solution if not stored appropriately;
Lipophilic properties necessitate careful solvent selection;
Handling requires standard laboratory safety protocols, including PPE and fume hood usage.
Keywords
Altretamine, Hexamethylmelamine, HMM, alkylating antineoplastic agent, DNA crosslinking, cytotoxicity research, preclinical cancer research, chemoresistance studies, laboratory research chemical, high-purity antineoplastic agent
Shipping Guarantee
All shipments are handled using validated cold-chain or controlled-environment logistics to preserve compound integrity. Packages are sealed in moisture-proof and light-protective containers and shipped via express international couriers with tracking and insurance.
Trade Assurance
We guarantee product authenticity, verified ≥99% purity, and compliance with analytical standards such as HPLC and NMR. Each batch is accompanied by a Certificate of Analysis (CoA). Our trade assurance policy ensures replacement or refund for any deviation from listed specifications.
Payment Support
Flexible and secure global payment options are available, including PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). Transactions are protected by industry-standard encryption.
Disclaimer
All products are intended for laboratory and preclinical research use only. They are not drugs, medical devices, or diagnostics, and should not be administered to humans or animals. Researchers must follow institutional biosafety guidelines. The information provided is for scientific reference and does not imply therapeutic efficacy or regulatory approval.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
1. What is Altretamine used for in research?
It is used in preclinical studies for DNA crosslinking, cytotoxicity assays, and chemoresistance research in tumor models.
2. Is Altretamine safe for human use?
No, it is intended strictly for laboratory and preclinical research.
3. How should Altretamine be stored?
Store at 2–8°C, protected from light, and avoid prolonged exposure to moisture.
4. Can it be dissolved in water for experiments?
Altretamine is poorly soluble in water; it is typically dissolved in DMSO or ethanol for laboratory use.
5. What is the purity of this compound?
≥99%, verified by HPLC and NMR.
6. Is a Certificate of Analysis included?
Yes, each batch includes a CoA confirming purity and analytical data.
7. Can Altretamine be combined with other chemotherapeutics in experiments?
Yes, commonly with platinum-based agents or other alkylating compounds for synergy studies.
8. What is the molecular weight of Altretamine?
237.37 g/mol.
9. Are there any experimental precautions?
Use PPE, handle in a fume hood, and avoid direct contact. Concentration should be titrated for cell studies.
10. What types of cancer models is Altretamine used in?
Primarily ovarian carcinoma, melanoma, and other solid tumor preclinical models.
11. Can it be used for in vitro cell cycle studies?
Yes, it induces G2/M arrest useful for cell cycle and DNA damage pathway investigations.
12. Is Altretamine lipophilic or hydrophilic?
It is lipophilic, requiring suitable organic solvents for dissolution.
13. How long is the shelf life?
24 months under recommended storage conditions.
14. What kind of experimental outcomes can be measured?
DNA crosslinks, apoptotic markers, cell viability, proliferation, and chemoresistance metrics.
15. Who supplies Altretamine?
Certified laboratory research chemical suppliers providing high-purity compounds with quality documentation.










Reviews
There are no reviews yet.