No products in the cart.
Sale
Dacarbazine Injection | CAS 4342-03-4 | Triazene Alkylating Agent for Melanoma and Sarcoma Research
Original price was: $3.00.$2.00Current price is: $2.00.
Dacarbazine (CAS 4342-03-4) is a synthetic triazene derivative widely used in oncology research as an alkylating agent. It plays a key role in studies investigating DNA methylation, tumor cell apoptosis, and mechanisms of chemotherapeutic resistance in melanoma and soft tissue sarcomas.
Description
Product Description
Dacarbazine, chemically known as 5-(3,3-dimethyl-1-triazeno)imidazole-4-carboxamide, is a synthetic alkylating agent belonging to the triazene class of compounds. It has been extensively studied in oncology research for its cytotoxic properties against rapidly dividing tumor cells, particularly in melanoma, Hodgkin’s lymphoma, and soft tissue sarcomas. Its chemical stability, metabolic activation pathways, and well-documented mechanism of DNA methylation make it a cornerstone molecule in experimental cancer pharmacology.
Chemical and Structural Overview
Dacarbazine has the molecular formula C6H10N6O and a molecular weight of 182.18 g/mol. It appears as a white to off-white crystalline powder, soluble in water and ethanol. The compound contains a triazene functional group that is critical to its alkylating capacity after metabolic activation in the liver. Upon activation, Dacarbazine generates diazomethane intermediates capable of methylating DNA at the O6 and N7 positions of guanine residues, leading to disruption of DNA replication and triggering apoptosis in cancer cells.
Research Significance
In research settings, Dacarbazine serves as a prototype compound for studying alkylating agents’ biochemical impact on nucleic acid integrity and tumor cell viability. It remains an important reference molecule in drug development programs aiming to design analogs with improved stability, reduced toxicity, and targeted delivery. Researchers also employ Dacarbazine in combination studies with interferon-alpha, cisplatin, or doxorubicin to explore synergistic antitumor effects and resistance mechanisms.
Physicochemical Properties
Appearance: White crystalline powder
Solubility: Soluble in water and slightly soluble in ethanol
Stability: Stable under dry, cool conditions but light sensitive
Melting Point: 220–224°C
Storage: -20°C, protected from light and moisture
Biological Research Applications
Dacarbazine’s biological activity is primarily mediated through alkylation of DNA bases, leading to mispairing and strand breaks. Its research applications span:
Cancer Pharmacology: Understanding DNA alkylation-induced apoptosis and mutagenesis
Chemoresistance Studies: Exploring cellular detoxification and DNA repair mechanisms in tumor cells
Drug Synergy Testing: Combining with other cytotoxic compounds for enhanced efficacy modeling
Biochemical Toxicology: Investigating oxidative stress and mitochondrial dysfunction following alkylating damage
Experimental Insights
In vitro, Dacarbazine demonstrates dose-dependent cytotoxicity across a range of melanoma and sarcoma cell lines. In vivo studies using murine xenograft models have revealed tumor regression correlated with increased DNA methylation damage. Furthermore, ongoing academic research utilizes Dacarbazine as a comparator in the testing of novel triazene analogs, allowing the evaluation of structure-activity relationships and pharmacodynamic optimization.
Product Specifications
| Item | Specification |
|---|---|
| Product Name | Dacarbazine |
| CAS Number | 4342-03-4 |
| Molecular Formula | C6H10N6O |
| Molecular Weight | 182.18 g/mol |
| Synonyms | DTIC, Imidazole carboxamide, DTIC-Dome |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99% |
| Solubility | Soluble in water; slightly soluble in ethanol |
| Storage Conditions | Store at -20°C, protected from light |
| Category | Triazene alkylating agent |
| Applications | Cancer research, DNA methylation studies, chemoresistance models |
| Research Area | Oncology, molecular pharmacology |
| Supplier Type | Research chemical manufacturer |
| Intended Use | For laboratory research use only |
Mechanism of Action
Dacarbazine’s mechanism of action involves metabolic activation by hepatic microsomal enzymes, particularly cytochrome P450 isozymes. The compound itself is a prodrug, requiring N-demethylation in the liver to form the active metabolite — 5-aminoimidazole-4-carboxamide (AIC) and diazomethane derivatives. The diazomethane species serve as potent methylating agents that covalently modify DNA.
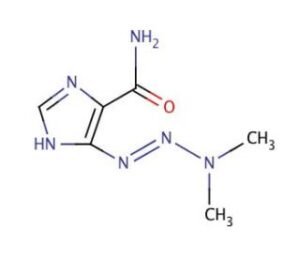
Stepwise Mechanistic Pathway
Metabolic Activation:
Dacarbazine undergoes N-demethylation via CYP1A1, CYP1A2, and CYP2E1 enzymes to yield the active diazomethane intermediate.DNA Methylation:
The diazomethane species methylate the O6 and N7 positions of guanine bases in DNA. This modification disrupts Watson-Crick base pairing, resulting in DNA strand mispairing and replication inhibition.DNA Repair and Cellular Response:
Alkylated DNA triggers the activation of DNA repair pathways such as MGMT (O6-methylguanine-DNA methyltransferase). When repair capacity is overwhelmed, apoptosis is induced via p53-mediated signaling cascades.Cytotoxic Outcome:
The culmination of DNA damage, repair exhaustion, and oxidative stress leads to apoptotic or necrotic cell death, depending on cell type and exposure duration.
Molecular Insights in Research
Dacarbazine is often used in studies evaluating:
The interplay between DNA methylation damage and cellular repair fidelity.
Epigenetic modifications associated with chemotherapeutic exposure.
The biochemical pathways underlying drug-induced apoptosis and oxidative stress.
Recent research emphasizes the role of Dacarbazine in stimulating endoplasmic reticulum stress and mitochondrial depolarization, offering valuable models for studying metabolic cytotoxicity and proteotoxic stress responses in tumor biology.
Synergistic Interactions
Dacarbazine demonstrates synergistic cytotoxic effects when combined with:
Interferon-α: Enhances immune-mediated tumor suppression.
Cisplatin or Doxorubicin: Augments DNA crosslinking and alkylation damage.
Temozolomide analogs: Provides insight into additive methylation mechanisms.
Such combination studies are essential in oncology research for identifying multidrug resistance mechanisms and improving chemotherapeutic index modeling.
Side Effects
Though not used clinically here, understanding Dacarbazine’s adverse effect profile remains critical for safe laboratory handling and accurate interpretation of research data.
Toxicological Profile in Research Models
Hepatotoxicity: Liver enzyme elevation due to metabolic activation stress.
Myelosuppression: Dose-dependent suppression of bone marrow activity observed in experimental animal models.
Gastrointestinal Irritation: Nausea, vomiting, and mucosal irritation reported in toxicity assays.
Neurotoxicity: At higher doses, neurological dysfunction has been documented in rodent studies.
Photosensitivity: Exposure to light can enhance degradation and increase phototoxic risk.
Safety Guidelines for Research Use
Handle in a biosafety cabinet (BSC) under appropriate ventilation.
Use nitrile gloves, lab coat, and protective eyewear.
Dispose of waste in accordance with hazardous chemical disposal protocols.
Avoid inhalation, ingestion, or contact with skin and mucous membranes.
Long-Term Exposure Observations
Chronic exposure models have shown oxidative DNA damage, mitochondrial swelling, and increased lipid peroxidation in hepatic tissues. Researchers are advised to use Dacarbazine strictly under approved laboratory containment procedures.
Keywords
Dacarbazine, CAS 4342-03-4, alkylating agent, triazene compound, DNA methylation, melanoma research, sarcoma research, apoptosis induction, chemoresistance, oncology research chemical, high purity peptide supplier, China peptide manufacturer, OEM peptide production, bulk peptide supplier.
Shipping Guarantee
All Dacarbazine shipments are handled using validated cold-chain logistics to preserve compound integrity. Each package is sealed in moisture-proof containers with secondary protective wrapping and continuous temperature monitoring. Products are shipped via express international couriers with full tracking and insurance coverage.
Trade Assurance
We Dacarbazine ensure product authenticity, verified ≥99% purity, and compliance with analytical standards (HPLC, MS, and NMR). Each batch is supplied with a Certificate of Analysis (CoA). Our trade assurance policy guarantees replacement or refund for any deviation from listed specifications.
Payment Support
We Dacarbazine provide flexible and secure global payment options to support international research transactions. Accepted payment methods include PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). All transactions are protected by industry-standard encryption and verified payment gateways to ensure confidentiality and fund security.
Disclaimer
This product is for laboratory research use only. It is not intended for human or veterinary use. Proper laboratory safety procedures should be observed when handling Dacarbazine. The information provided is for educational and research purposes only.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
1. What is the primary research use of Dacarbazine?
Dacarbazine is used to study DNA methylation, apoptosis, and mechanisms of chemotherapeutic resistance in cancer models.
2. What purity level is provided for Dacarbazine?
All Dacarbazine batches are verified to have a purity of ≥99% as confirmed by HPLC and MS analysis.
3. Can Dacarbazine be used for clinical trials?
No, it is strictly intended for laboratory research use only, not for human administration.
4. How should Dacarbazine be stored?
It should be stored at -20°C, protected from light and moisture to maintain chemical stability.
5. What type of solvent is suitable for Dacarbazine dissolution?
Dacarbazine dissolves well in sterile water and is slightly soluble in ethanol.
6. Does Dacarbazine require metabolic activation in studies?
Yes, it acts as a prodrug and must be metabolically activated by liver enzymes in experimental systems.
7. Is Dacarbazine available in bulk quantity?
Yes, we provide OEM and bulk supply options for research institutions and pharmaceutical developers.
8. What analytical methods confirm Dacarbazine purity?
Purity is verified through HPLC, MS, and NMR according to analytical quality standards.
9. How is Dacarbazine shipped internationally?
It is shipped in insulated, moisture-proof containers via express couriers under temperature-controlled logistics.
10. Is Dacarbazine classified as hazardous?
Yes, as an alkylating compound, it should be handled with care under laboratory safety protocols.
11. What are the major research areas using Dacarbazine?
It is widely used in oncology, pharmacology, and molecular biology research involving DNA alkylation.
12. Do you offer documentation for research compliance?
Yes, each shipment includes a Certificate of Analysis (CoA) for verification.
13. Can Dacarbazine be customized for formulation studies?
Yes, we can provide customized concentrations and formulations for specific research needs.
14. Is this product sourced from a China peptide manufacturer?
Yes, we are a verified China-based research chemical and peptide supplier offering global shipping.
15. Why choose your factory for peptide and small molecule supply?
We are a factory peptide supplier specializing in high-purity compounds, competitive pricing, and OEM bulk peptide production services.








Reviews
There are no reviews yet.