No products in the cart.
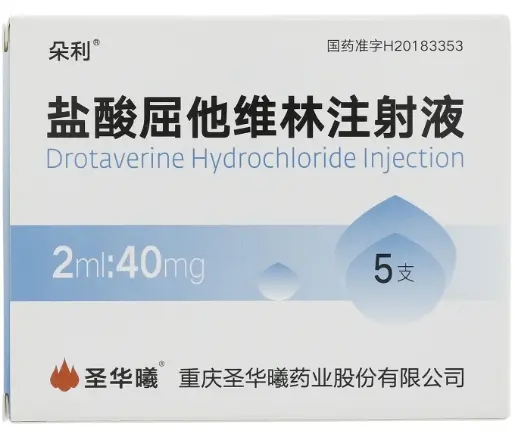
Drotaverine Hydrochloride 40?mg ×?5 Vials Wholesale price comparison
$2.00
Drotaverine is a spasmolytic PDE4 inhibitor used in preclinical research to relieve smooth muscle spasms—gastrointestinal, biliary, urinary, and uterine. Each vial contains 40?mg in 2?mL solution. Manufactured by Chongqing Shenghuaxi Pharmaceutical Co., Ltd., approved under NMPA H20183353, product code 86909568000197, barcode 6933982500616.
?? For laboratory research use only.?Please consult the staff for other purposes?
Description
Product Specifications
| Attribute | Details |
|---|---|
| Product Name | Drotaverine Hydrochloride Injection |
| Generic Name | Drotaverine Hydrochloride |
| CAS Number | 985?12?6 |
| Molecular Formula | C??H??NO? · HCl |
| Molecular Weight | ~433.97?g/mol |
| Dosage Form | Solution for IM/IV injection |
| Strength | 40?mg per 2?mL vial |
| Pack Size | 5 vials per box |
| Approval No. | H20183353 (China NMPA) |
| Product Code | 86909568000197 |
| Manufacturer | Chongqing Shenghuaxi Pharmaceutical Co., Ltd. |
| Barcode | 6933982500616 |
| Storage | Store at 2–8?°C, protect from light |
| Intended Use | Laboratory research use only |
Mechanism of Action
Drotaverine selectively inhibits phosphodiesterase?4 (PDE4) and blocks L-type calcium channels, leading to smooth muscle relaxation without anticholinergic side effects. Useful in studies involving colic, dysmenorrhea, and uterine dilation.
Research Applications & Pharmacology
Smooth muscle spasm models: Effective in gastrointestinal, genitourinary, biliary, and uterine muscle studies (e.g., labor augmentation, renal colic relief)?go.drugbank.com.
Pharmacokinetics: High bioavailability (~65%), peak effect in 45–60 min, long half-life (7–16?hrs), 95–98% protein bound, hepatic elimination, urinary and biliary excretion?medchemexpress.com+2en.wikipedia.org+2clinicaltrials.gov+2.
Safety Profile & Handling
Adverse Effects:
Possible dizziness, headache, flushing, hypotension, tachycardia, rare arrhythmia/anaphylaxis.
Overdose can lead to AV block, respiratory depression, seizures, cardiac arrest.
Handling Guidelines:
Use PPE, aseptic technique.
Administer slowly via IM/IV; aspirate to avoid intravascular injection.
Monitor animals for cardiovascular and CNS responses.
Dispose of vials as pharmaceutical waste.
?? Research?Use Disclaimer
This product is intended solely for laboratory research and not approved for diagnosis, therapy, clinical, or veterinary use. Misuse may cause safety hazards and compromise scientific data.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |








Reviews
There are no reviews yet.