No products in the cart.
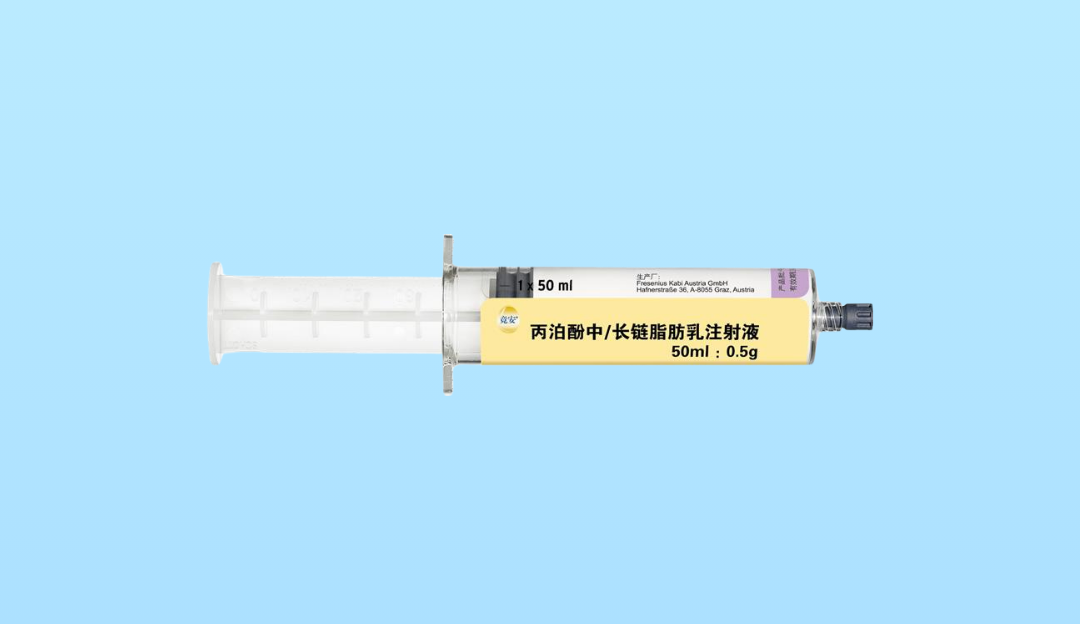
Propofol 20?mg/mL MCT/LCT Emulsion Injection 20?mL ×?5 Vials
$1.00
This propofol formulation uses a medium-chain and long-chain triglyceride (MCT/LCT) emulsion, designed to produce equivalent anesthetic effects compared to long-chain-only emulsions while reducing injection pain and minimizing triglyceride accumulation. Each 20?mL vial contains 400?mg propofol (20?mg/mL).
?? For laboratory research use only?Please consult staff for other specifications and uses?
Description
Product Specifications
| Attribute | Details |
|---|---|
| Product Name | Propofol MCT/LCT Fat Emulsion Injection |
| Generic Name | Propofol (2,6?diisopropylphenol) |
| CAS Number | 2078?54?8 zh.wikipedia.org+4en.wikipedia.org+4zh.wikipedia.org+4 |
| Molecular Formula | C??H??O |
| Molecular Weight | ~178.27?g/mol |
| Dosage Form | Sterile lipid emulsion for IV injection |
| Strength | 20?mg/mL (400?mg per 20?mL vial) |
| Pack Size | 5 vials per box |
| Approval Number | H20203504 (China NMPA) |
| Product Code | 86910137000029 |
| Manufacturer | Jiangsu Yingke Biopharma Co., Ltd. |
| Barcode | 6970465112126 |
| Storage | Store at 2–25?°C; protect from light and freezing |
| Intended Use | Laboratory research only |
Mechanism of Action
Propofol acts as a GABA? receptor agonist, inducing CNS depression and anesthesia. Using an MCT/LCT emulsion maintains pharmacodynamics while reducing free propofol concentration, decreasing injection pain and triglyceride load.
Research Applications & Pharmacology
Sedation & anesthesia research: Supports induction and maintenance models using mechanical ventilation or neuromuscular blockade .
Comparison studies: MCT/LCT emulsions exhibit similar efficacy to LCT emulsions but show faster triglyceride clearance and less injection pain.
Pharmacokinetics: Rapid onset (~15–30?s), short duration (~5–10?min), high protein binding (95–99%), metabolized hepatically with variable elimination half-life (1.5–24?hr).
Safety Profile & Handling
Side effects: Hypotension, injection-site pain, respiratory depression, risk of propofol infusion syndrome during prolonged use.
Handling guidance: Administer under sterile conditions; monitor respiratory and cardiovascular parameters closely; dispose of residual lipid emulsions per hazardous waste regulations.
?? Research?Use Disclaimer
This product is intended solely for laboratory research. It is not approved for any clinical, diagnostic, therapeutic, or veterinary use. Improper use may result in serious safety risks and compromise experimental validity.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |





Reviews
There are no reviews yet.