No products in the cart.
OSSOTIDE Injection (GUSHU) – 10mg – Low price wholesale comparison
$1.00
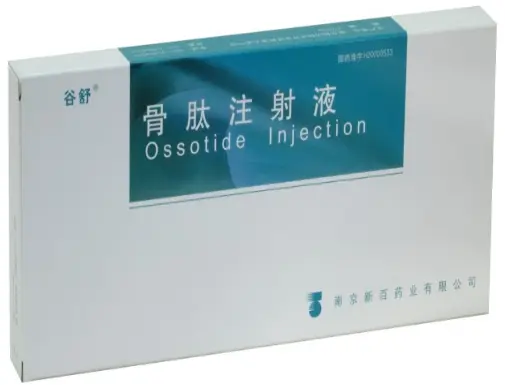
OSSOTIDE Injection (GUSHU) is a clinical-grade injectable bone peptide formulation manufactured by Nanjing Xinbai Pharmaceutical Co., Ltd. Each vial delivers 10mg/2ml of active peptide for supporting bone tissue regeneration and repair. Supplied in a box of 10 vials. NMPA-approved.?Please consult staff for price, other specifications and uses?
Description
OSSOTIDE Injection (GUSHU) is a sterile injectable bone peptide preparation developed to assist in the treatment of orthopedic conditions such as fractures, osteoporosis, and degenerative joint diseases. Its mechanism involves stimulating osteoblast proliferation, promoting collagen synthesis, and enhancing calcium deposition in the bone matrix.
Each 2ml vial contains 10mg of purified bone-derived polypeptides. OSSOTIDE has been widely adopted in hospitals and clinics throughout China and is backed by both in vivo and clinical research.
Manufactured by Nanjing Xinbai Pharmaceutical Co., Ltd., the formulation is certified under Chinese NMPA approval H20003533 and is distributed in tamper-proof boxes of 10 vials.
?? This product is approved for clinical use in China. Please consult your country’s local regulatory framework before use.
Product Specifications
| Parameter | Details |
|---|---|
| Product Name | OSSOTIDE Injection (GUSHU) |
| Generic Name | Bone Peptide Injection |
| CAS Number | N/A (natural peptide complex) |
| Molecular Formula | Peptide mixture (variable) |
| Molecular Weight | Variable |
| Dosage Form | Injection |
| Strength | 10mg per 2ml vial |
| Quantity | 10 vials per box |
| Approval Number | H20003533 (China NMPA) |
| Product Code | 86901598000284 |
| Barcode | 6913614006204 |
| Manufacturer | Nanjing Xinbai Pharmaceutical Co., Ltd. |
| Indications | Fractures, osteoporosis, osteoarthritis, bone loss |
| Usage | Intramuscular/intravenous injection by licensed provider |
| Storage Conditions | Refrigerate at 2–8°C, avoid light exposure |
Mechanism & Clinical Applications
OSSOTIDE Injection acts by:
Stimulating osteoblast differentiation
Enhancing synthesis of Type I collagen
Promoting calcium and phosphate mineralization
Speeding up healing of fractures and bone defects
Clinical Applications:
Osteoporosis (post-menopausal or age-related)
Non-union or delayed-union fractures
Osteoarthritis and joint degeneration
Post-surgical bone recovery
Spinal fusion, bone graft integration
Known Reactions (Clinical Setting)
In human use under physician guidance, OSSOTIDE has been associated with:
Mild localized pain at the injection site
Rare allergic skin reactions
Transient serum calcium changes (monitoring recommended)
Always follow local clinical protocol and consult a healthcare provider.
Safety & Handling Guidelines
Prescription Required: Yes
Administration: Intramuscular or intravenous only under medical supervision
Storage: Refrigerate 2–8°C
Disposal: Treat as biohazard material according to local regulations
Core Keywords
ossotide injection
GUSHU bone peptide
ossotide 10mg 2ml
fracture healing peptide
osteoporosis injectable peptide
Nanjing Xinbai ossotide
H20003533 ossotide
bone regeneration injection
bone matrix peptide
osteoblast peptide treatment
Packaging & Supply Notes
Package Configuration: 10 single-dose vials per box
Shipping Compliance: Cold-chain shipping supported
Customs & Regulations: Buyer is responsible for compliance with import regulations. Contact us for assistance.
Regulatory Notice
This product is approved for human clinical use in China. Outside China, it may be subject to regulatory limitations. It is not FDA-approved for clinical use in the US or EU. Ensure compliance with your local health authority before purchase or administration.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
Q1: Is OSSOTIDE Injection approved for clinical use?
A1: Yes. It is approved by the Chinese National Medical Products Administration (NMPA) under registration H20003533 for clinical use.
Q2: What’s the recommended dosage?
A2: Typical administration involves 1 vial (10mg/2ml) per day or every other day for 2–4 weeks. Exact dosing must be prescribed by a physician.
Q3: Can I order OSSOTIDE in bulk for my clinic or pharmacy?
A3: Absolutely. Bulk and wholesale quantities are available with international shipping options. Contact us for volume pricing.
Q4: Are supporting documents available?
A4: Yes. COA (Certificate of Analysis), product insert, and regulatory documentation available upon request.
Q5: What are the storage requirements?
A5: OSSOTIDE must be stored refrigerated between 2–8°C and protected from light to preserve peptide stability.





Reviews
There are no reviews yet.