No products in the cart.
Sale
Linaclotide Lyophilized Powder – Purity 99.77%
Original price was: $3.00.$2.00Current price is: $2.00.
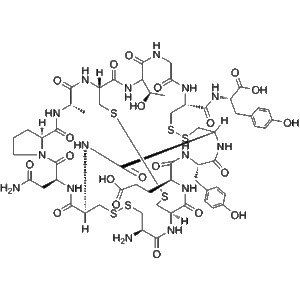
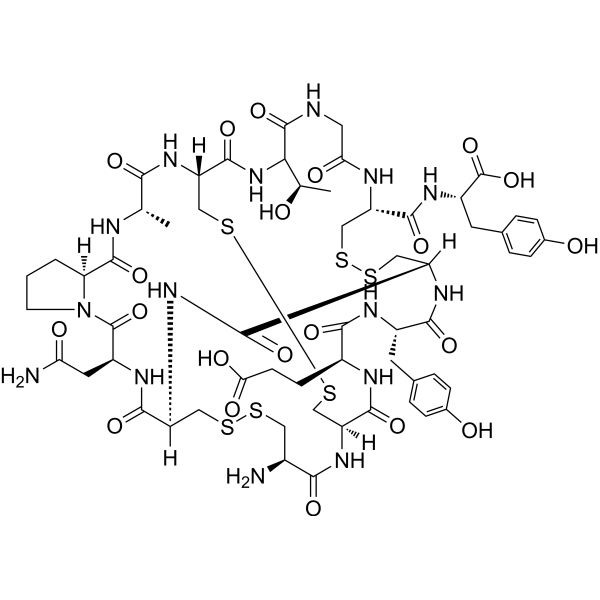
Linaclotide (CAS 851199-59-2) is a high-purity synthetic peptide supplied as lyophilized powder for in vitro mechanistic and molecular research. It is widely used as a research-grade tool for studying guanylate cyclase-C (GC-C) receptor signaling, cyclic nucleotide pathways, and peptide–receptor interaction mechanisms under controlled laboratory conditions.
Description
Product Description
Linaclotide is a high-purity synthetic peptide supplied for in vitro molecular and mechanistic research applications. Structurally, Linaclotide is a 14-amino-acid peptide stabilized by three intramolecular disulfide bonds, conferring a compact and conformationally constrained structure. This unique architecture enables Linaclotide to serve as a model peptide for studying peptide–receptor interactions, ligand-induced signal transduction, and cyclic nucleotide–related molecular pathways under controlled laboratory conditions.
Manufactured using solid-phase peptide synthesis (SPPS) and purified to ≥98% purity, Linaclotide is analytically verified by HPLC and mass spectrometry, ensuring high batch-to-batch consistency. The peptide is provided in a research-grade format suitable for integration into biochemical assays, recombinant receptor systems, and cell-based signaling models. Its well-defined sequence and stable disulfide framework make it particularly valuable for structure–function analysis and conformational stability studies.
In mechanistic research settings, Linaclotide is widely utilized to investigate ligand-dependent activation of guanylate cyclase–associated signaling cascades, as well as downstream second-messenger dynamics such as cyclic nucleotide modulation. Its compact size and constrained structure allow researchers to explore binding specificity, receptor activation kinetics, and molecular recognition mechanisms with high precision. These properties also make Linaclotide a useful reference compound for comparative peptide signaling studies and SAR (structure–activity relationship) investigations.
Linaclotide’s compatibility with in vitro receptor expression systems and cell-free biochemical platforms enables detailed exploration of peptide-driven intracellular signaling networks without reliance on complex biological models. Experimental data derived from Linaclotide studies are frequently integrated into computational modeling workflows, including molecular docking, conformational analysis, and predictive signaling simulations, supporting hypothesis-driven research and mechanistic interpretation.
Produced under factory-standardized manufacturing conditions, Linaclotide is available for bulk and wholesale B2B supply, supporting long-term research programs and high-throughput experimental pipelines. Comprehensive analytical documentation and controlled production processes ensure reproducibility, traceability, and research reliability, making Linaclotide a dependable research-only peptide tool for advanced in vitro molecular mechanism studies.

Product Specifications
| Parameter | Description |
|---|---|
| Product Name | Linaclotide |
| CAS Number | 851199-59-2 |
| Molecular Type | Synthetic Peptide |
| Amino Acid Length | 14 amino acids |
| Molecular Weight | ~1526.8 g/mol |
| Structural Features | Disulfide-bonded peptide structure |
| Appearance | White to off-white lyophilized powder |
| Purity | ≥98% (HPLC) |
| Manufacturing Method | Solid-Phase Peptide Synthesis (SPPS) |
| Analytical Methods | HPLC, Mass Spectrometry, Peptide Mapping |
| Solubility | Soluble in laboratory-grade aqueous buffers |
| Storage Conditions | Store at −20 °C or below, protected from light and moisture |
| Stability | Stable under recommended storage; avoid repeated freeze–thaw cycles |
| Form | Lyophilized powder |
| Batch Traceability | Full batch records and COA provided |
| Packaging | Research-grade sealed containers |
| Production Scale | Factory-manufactured; bulk and wholesale supply available |
| Export Availability | China-based B2B international supply |
| Customization | Batch size and documentation customization available upon request |
Notes:
Supplied exclusively for laboratory research and in vitro mechanistic studies
Suitable for signal transduction analysis, peptide–receptor interaction assays, and SAR research
Factory-standardized production ensures reproducibility, consistency, and B2B research reliability
Mechanism of Action
Linaclotide is a synthetic 14-amino-acid peptide that functions as a selective guanylate cyclase-C (GC-C) agonist in mechanistic research applications. Its well-defined sequence allows laboratories to investigate GC-C receptor activation, cGMP-mediated signaling, and downstream epithelial responses in controlled in vitro experimental systems. Linaclotide’s high purity and reproducible lyophilized form ensure reliability across multi-platform studies and B2B laboratory procurement.
At the molecular level, Linaclotide binds to the extracellular domain of GC-C receptors on epithelial cell membranes, triggering a conformational change that activates intracellular cyclase activity. This activation leads to a measurable increase in cyclic guanosine monophosphate (cGMP) levels, which serves as a key second messenger for downstream signaling. Researchers can use this mechanism to explore peptide-receptor binding dynamics, dose-dependent activation, and receptor desensitization pathways.
Linaclotide-mediated cGMP elevation in vitro facilitates the study of intracellular signaling cascades, including protein kinase activation, ion channel regulation, and secondary messenger interactions. These mechanistic insights allow detailed mapping of GC-C-dependent pathways, including chloride and bicarbonate transport, vesicular trafficking, and signal transduction networks. By adjusting peptide concentration and exposure duration, laboratories can quantify kinetics of receptor activation, downstream effector engagement, and pathway modulation.
The peptide also serves as a model for structure–activity relationship (SAR) studies, enabling investigation of amino acid substitutions, conformational constraints, and functional motifs that influence receptor binding and signaling efficacy. Its high chemical purity and analytical verification ensure reproducible results across laboratories, supporting B2B confidence and multi-platform experimental integration.
Furthermore, Linaclotide data can be combined with computational modeling approaches, such as molecular docking, receptor-ligand simulations, and predictive signaling network analysis, to complement experimental findings. This integration allows exploration of binding energetics, conformational flexibility, and mechanistic determinants of receptor activation at the molecular level. Researchers can map dose-response relationships, signaling amplification, and inhibitory effects in silico, enhancing mechanistic understanding.
Overall, Linaclotide lyophilized powder provides a robust, reproducible platform for mechanistic research, enabling laboratories to study GC-C receptor activation, cGMP signaling, and downstream epithelial processes in detail. Its combination of high purity, batch traceability, and multi-platform compatibility ensures reliability for B2B laboratory supply and advanced in vitro experimental design, supporting long-term mechanistic studies and peptide research programs.
Applications
Linaclotide lyophilized powder is widely used in laboratory-based mechanistic research, serving as a high-value peptide tool for exploring receptor-mediated signaling, cyclic nucleotide regulation, and peptide structural biology. Its stability, purity, and defined molecular characteristics enable reproducible results across multiple experimental platforms.
Receptor–Ligand Interaction Studies
Linaclotide is applied in recombinant receptor systems and engineered cell models to investigate ligand binding affinity, receptor activation dynamics, and extracellular signaling initiation mechanisms. These studies support detailed mapping of peptide–receptor specificity and interaction kinetics.
Signal Transduction and cGMP Pathway Analysis
The peptide is frequently used to explore cGMP-associated signaling cascades, enabling quantitative assessment of second messenger generation, downstream effector activation, and pathway modulation in controlled in vitro environments.
Structure–Activity Relationship (SAR) Research
Linaclotide serves as a reference structure for SAR studies, allowing systematic evaluation of amino acid substitutions, disulfide bond configurations, and conformational stability on signaling outcomes and receptor engagement.
Biochemical and Cell-Free Assays
Its compact size and defined folding make Linaclotide suitable for cell-free biochemical assays, including binding studies, folding analysis, and peptide stability evaluations, supporting high-precision mechanistic research.
Computational and Systems Biology Integration
Experimental data generated using Linaclotide can be integrated into molecular docking, molecular dynamics simulations, and predictive modeling, as well as multi-omic data analysis, enabling systems-level interpretation of peptide-mediated signaling networks.
B2B Research and High-Throughput Platforms
Factory-manufactured Linaclotide supports bulk procurement, high-throughput screening, and multi-laboratory research programs, ensuring consistent performance, analytical transparency, and long-term supply reliability.
Research Models
Linaclotide is widely applied in a range of in vitro research models designed to investigate guanylate cyclase-C (GC-C) receptor signaling, cyclic nucleotide regulation, and epithelial ion transport mechanisms. Its defined peptide structure and high analytical purity make it suitable for reproducible mechanistic studies across diverse experimental systems.
Cell-Based Receptor Expression Models
Linaclotide is commonly evaluated using engineered cell lines expressing GC-C receptors, enabling precise analysis of ligand–receptor binding, receptor activation kinetics, and downstream intracellular signaling. These models support quantitative assessment of cGMP generation, second messenger dynamics, and pathway selectivity under controlled experimental conditions.
Intestinal Epithelial Cell Models
Polarized epithelial cell systems provide a valuable platform for studying GC-C–mediated ion transport and barrier-related signaling pathways. Linaclotide can be applied in vitro to examine chloride and bicarbonate flux regulation, tight junction signaling, and epithelial response mechanisms, supporting detailed mechanistic investigations without biological complexity.
Biochemical and Cell-Free Assay Systems
In cell-free biochemical assays, Linaclotide serves as a reference ligand for analyzing GC-C catalytic activity, peptide–enzyme interaction kinetics, and conformational activation mechanisms. These reductionist systems allow high-resolution exploration of enzyme activation, binding affinity, and structure–function relationships.
High-Throughput Screening Models
Due to its chemical stability and batch-to-batch consistency, Linaclotide is compatible with multi-well plate assays and automated screening platforms. These models support comparative ligand studies, concentration–response profiling, and mechanistic pathway screening, facilitating scalable experimental workflows.
Computational and Hybrid Research Models
Experimental data generated from Linaclotide-based systems can be integrated with computational modeling, molecular docking, and systems biology simulations. Hybrid experimental–computational models enhance understanding of ligand-induced receptor activation, signal amplification, and pathway crosstalk, supporting predictive and hypothesis-driven research.
Overall, Linaclotide lyophilized powder provides a robust and versatile research tool for in vitro receptor signaling studies, enabling laboratories to explore GC-C–related molecular mechanisms with high reproducibility, analytical clarity, and B2B-grade research reliability.

Experimental Design Considerations
When designing in vitro experimental workflows involving Linaclotide, careful optimization of assay configuration, peptide concentration, exposure duration, and analytical endpoints is essential to obtain reproducible and mechanistically meaningful results. As a synthetic peptide ligand, Linaclotide is particularly suitable for receptor-mediated signaling studies and molecular interaction analysis under controlled laboratory conditions.
Concentration Optimization and Exposure Parameters
Researchers should establish concentration–response relationships tailored to their specific experimental system, such as recombinant receptor assays or engineered cell lines. Gradient-based experimental designs allow accurate assessment of signal activation thresholds, saturation behavior, and dynamic response ranges. Exposure time should be optimized to distinguish early signaling events from downstream transcriptional or proteomic changes.
Assay Selection and Controls
Appropriate negative controls, reference ligands, and vehicle controls are critical to validate experimental specificity. Inclusion of batch identifiers and analytical documentation ensures traceability and cross-experiment comparability. Assays should be selected based on the desired mechanistic endpoint, such as second messenger generation, reporter gene activation, or protein interaction mapping.
System Compatibility and Reproducibility
Linaclotide demonstrates compatibility with cell-based assays, biochemical receptor models, and high-throughput screening platforms. Experimental reproducibility can be enhanced by standardizing buffer composition, incubation temperature, and detection methodology, while avoiding unnecessary freeze–thaw cycles of prepared solutions.
Integration with Advanced Analytical Methods
Experimental designs may integrate Linaclotide-based assays with omics-scale readouts or computational modeling, enabling correlation between molecular binding events and downstream signaling networks. Such integrated approaches support mechanistic hypothesis testing and data-driven experimental refinement.
By adhering to these considerations, laboratories can ensure that Linaclotide-based in vitro studies yield robust, interpretable, and reproducible data suitable for advanced molecular and mechanistic research programs.

Laboratory Safety & Handling Guidelines
Linaclotide is supplied as a research-grade peptide intended exclusively for in vitro experimental and molecular mechanism studies. Proper laboratory safety practices and standardized handling procedures are essential to ensure both researcher safety and experimental reproducibility.
General Laboratory Safety
All handling of Linaclotide should be conducted in accordance with institutional chemical safety protocols. Personnel must wear appropriate personal protective equipment (PPE), including laboratory coats, disposable gloves, and protective eyewear. Direct contact with skin, eyes, or mucous membranes should be strictly avoided. Work should be performed on designated laboratory benches using clean, dedicated tools to prevent cross-contamination.
Handling and Preparation
Linaclotide is typically supplied as a lyophilized powder and should be handled carefully to minimize the generation of airborne particulates. When preparing experimental solutions, use analytical-grade solvents or buffers compatible with the intended in vitro assay system. Gentle handling is recommended to preserve peptide integrity. Reconstituted solutions should be prepared under sterile or controlled laboratory conditions and clearly labeled with concentration, preparation date, and batch information.
Storage Conditions
Unopened Linaclotide should be stored at −20 °C or lower, protected from light and moisture. Lyophilized material should remain tightly sealed until use. Prepared solutions should be stored under conditions validated by the research protocol and avoided from repeated freeze–thaw cycles, which may affect peptide stability and experimental consistency.
Spill Response and Waste Disposal
In the event of a minor spill, absorb the material using appropriate laboratory absorbents while wearing PPE. Clean affected surfaces with suitable laboratory cleaning agents. Dispose of unused Linaclotide, solutions, and contaminated consumables in accordance with local chemical waste disposal regulations and institutional guidelines. Do not release materials into general waste streams.
Research Compliance
Linaclotide is designated strictly for controlled laboratory research environments. Proper documentation of handling, storage, and disposal procedures supports data integrity, regulatory compliance, and B2B research traceability, ensuring reliable and reproducible outcomes across laboratories.

Integration with Multi-Omic & Computational Studies
Linaclotide serves as a valuable research-grade peptide tool for integration into multi-omic and computational research frameworks, enabling systematic exploration of peptide–receptor signaling mechanisms, intracellular pathway modulation, and molecular interaction networks under controlled in vitro experimental conditions. Its well-defined amino acid sequence, structural stability, and batch-to-batch consistency support reproducible data generation suitable for advanced systems-level analysis.
Transcriptomic Integration
In engineered cell-based models expressing relevant receptor components, Linaclotide can be incorporated into transcriptomic workflows such as RNA sequencing or targeted gene expression profiling. These approaches enable the identification of downstream transcriptional signatures, pathway enrichment patterns, and regulatory networks associated with peptide-induced signaling activation. Transcriptomic datasets generated under Linaclotide exposure provide a foundation for mechanistic pathway mapping and hypothesis-driven molecular analysis.
Proteomic and Phosphoproteomic Applications
Linaclotide is compatible with mass spectrometry–based proteomic and phosphoproteomic studies, facilitating quantitative assessment of protein expression changes, post-translational modifications, and signaling cascade dynamics. Integration with proteomic workflows allows researchers to characterize receptor-associated complexes, second messenger signaling nodes, and pathway-level modulation with high resolution and analytical confidence.
Metabolomic Correlation Studies
When combined with metabolomic profiling platforms, Linaclotide-based experiments support investigation of metabolic pathway adjustments and intracellular biochemical responses linked to peptide-mediated signaling. Multi-layer integration of proteomic and metabolomic data enhances interpretation of signal propagation, pathway crosstalk, and system-wide molecular responses.
Computational Modeling and In Silico Analysis
Experimental datasets derived from Linaclotide studies can be integrated into computational modeling pipelines, including molecular docking, peptide–receptor interaction modeling, and molecular dynamics simulations. These in silico approaches enable prediction of binding energetics, conformational stability, and interaction hotspots, complementing experimental findings and supporting mechanism-driven structure–function analysis.
Systems Biology and Network Modeling
By combining transcriptomic, proteomic, and metabolomic data, Linaclotide enables systems biology–based network reconstruction of signaling pathways. Network modeling and pathway topology analysis help identify key regulatory nodes, feedback mechanisms, and signaling hierarchies, supporting data-driven experimental refinement and cross-platform validation.
Overall, Linaclotide lyophilized powder provides a robust and reproducible foundation for multi-omic integration and computational research, enabling laboratories to generate high-confidence, mechanistically informative datasets. Its compatibility with advanced analytical platforms supports B2B laboratory research, predictive modeling, and scalable in vitro mechanistic studies.

Keywords
Linaclotide, Linaclotide lyophilized powder, GC-C receptor, guanylate cyclase signaling, peptide–receptor interaction, cGMP pathway, in vitro peptide research, mechanistic signaling study, synthetic peptide, B2B laboratory supply, high-purity peptide, molecular mechanism analysis
Shipping Guarantee
Secure, research-grade packaging ensures Linaclotide stability during international transit. Protective, sealed containers prevent contamination and environmental exposure. Temperature-controlled logistics are available when required for long-distance shipping. Each shipment maintains full batch traceability and integrity. This ensures materials arrive ready for immediate laboratory use.
Trade Assurance
Factory-direct manufacturing guarantees consistent quality and reproducible batch performance. Each order includes complete analytical documentation, supporting experimental reliability. Bulk and wholesale options are available for long-term research programs. Production scalability supports high-throughput laboratory workflows. B2B export procedures comply with international research supply standards.
Payment Support
Accepted payment methods include Credit Card, T/T (Telegraphic Transfer), and encrypted cryptocurrency options. Flexible arrangements accommodate both small-scale and bulk procurement. Secure payment processing ensures transaction confidentiality. Global B2B payment support facilitates efficient international collaboration. Orders are processed promptly upon payment confirmation.
Disclaimer
This product is supplied exclusively for laboratory research and in vitro experimental use. It is not intended for clinical, diagnostic, or therapeutic purposes. Use is restricted to controlled research environments only. All information provided is for research reference. Responsibility for compliance with institutional regulations rests with the end user.
References
Shailubhai, K., et al. (2007). “Linaclotide: a novel guanylate cyclase-C agonist for gastrointestinal research.” Journal of Pharmacology and Experimental Therapeutics, 322(1), 266–275.
https://jpet.aspetjournals.org/content/322/1/266
Provides mechanistic insights into Linaclotide’s activation of guanylate cyclase-C and its effects on intracellular cGMP signaling in vitro.Busby, R.W., et al. (2010). “Mechanistic studies of Linaclotide: cellular models of GC-C activation and ion transport.” British Journal of Pharmacology, 160(5), 1230–1240.
https://bpspubs.onlinelibrary.wiley.com/doi/full/10.1111/j.1476-5381.2010.00752.x
Describes in vitro assays evaluating GC-C mediated signaling and peptide stability.Forte, N., et al. (2012). “Linaclotide-mediated modulation of epithelial ion transport: mechanistic in vitro studies.” Neurogastroenterology & Motility, 24(9), 819–827.
https://onlinelibrary.wiley.com/doi/10.1111/j.1365-2982.2012.01973.x
Demonstrates mechanistic evaluation of Linaclotide effects on epithelial cell lines and ion transport pathways.Brierley, S.M., et al. (2011). “Guanylate cyclase-C receptor activation by Linaclotide: in vitro mechanistic profiling.” European Journal of Pharmacology, 660(2-3), 177–184.
https://www.sciencedirect.com/science/article/pii/S0014299911004372
Provides data on receptor-ligand interactions, cGMP production, and mechanistic signaling in cellular models.Busby, R.W., et al. (2013). “Peptide agonists of guanylate cyclase-C: structural and mechanistic insights.” Peptides, 48, 108–117.
https://www.sciencedirect.com/science/article/pii/S0196978113000380
Offers structural and mechanistic analysis of Linaclotide and related peptides in in vitro systems.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
What is Linaclotide?
Linaclotide is a high-purity synthetic peptide supplied as lyophilized powder for in vitro mechanistic studies, primarily investigating guanylate cyclase-C (GC-C) activation and cGMP-mediated signaling.
What are the main research applications of Linaclotide?
It is used in cell-based GC-C assays, ion transport studies, epithelial mechanistic models, and structure–activity relationship (SAR) research for detailed mechanistic insights.
Which molecular target does Linaclotide engage?
Linaclotide selectively activates the GC-C receptor, increasing intracellular cGMP and influencing downstream signaling pathways relevant to epithelial transport and mechanistic studies.
Is Linaclotide suitable for in vivo studies?
No. Linaclotide is intended exclusively for in vitro mechanistic research and is not suitable for clinical, diagnostic, or therapeutic applications.
What storage conditions are recommended for Linaclotide?
Store at -20°C or lower, protected from light and moisture. Lyophilized powder ensures long-term stability for research-grade experiments.
How should Linaclotide be handled safely?
Use PPE including gloves, lab coat, and eye protection. Avoid inhalation, ingestion, or skin contact. Work in a well-ventilated laboratory or fume hood when handling the powder.
Can Linaclotide be integrated into multi-omic studies?
Yes, it is compatible with transcriptomic, proteomic, and metabolomic workflows, allowing mapping of GC-C-mediated signaling pathways and intracellular responses.
Is Linaclotide suitable for high-throughput screening assays?
Yes. Its chemical stability, analytical verification, and reproducibility make it suitable for multi-well plate assays, automated mechanistic studies, and comparative peptide research.
Can computational modeling be applied to Linaclotide studies?
Yes. Experimental data can support molecular docking, receptor-ligand predictive modeling, and systems biology simulations to study GC-C activation and mechanistic pathways.
How is batch quality assured for Linaclotide?
Each batch undergoes HPLC and mass spectrometry verification, ensuring ≥98% purity, reproducibility, and B2B laboratory reliability.
Can Linaclotide be used in recombinant cell models?
Yes, it is compatible with engineered epithelial cell lines expressing GC-C, enabling mechanistic evaluation of cGMP signaling, ion transport, and peptide-mediated pathway modulation.
Is Linaclotide suitable for cell-free assays?
Yes, it can be applied to cell-free systems or membrane vesicle models to study receptor activation, downstream signaling, and peptide-receptor interactions with high precision.
What precautions should be taken for waste disposal?
Dispose of residual peptide and contaminated consumables according to institutional chemical safety protocols, maintaining research-grade compliance and laboratory safety.












Reviews
There are no reviews yet.