No products in the cart.
Sale
Enfuvirtide Lyophilized Powder | High Purity 98% | Anti-HIV Fusion Inhibitor
Original price was: $26.00.$23.00Current price is: $23.00.
Enfuvirtide (CAS 159519‑65‑0) is a high-purity synthetic peptide provided as lyophilized powder, intended exclusively for in vitro mechanistic and molecular studies. It serves as a research-grade tool for investigating fusion inhibition, peptide-receptor interactions, and signal transduction pathways in controlled laboratory settings.
Description
Contents
hide
Product Description
Enfuvirtide is a synthetic 36-amino-acid peptide designed to study viral fusion mechanisms and peptide-mediated inhibition of membrane interactions. Its well-characterized sequence allows researchers to examine molecular binding, conformational changes, and mechanistic pathways in in vitro experimental models.
Produced via solid-phase peptide synthesis (SPPS) under rigorous quality control, Enfuvirtide achieves ≥98% purity, verified by HPLC and mass spectrometry, ensuring reproducible results across laboratories. The lyophilized powder form facilitates long-term storage and integration into diverse assay systems, including cell-based fusion assays, recombinant protein models, and biochemical interaction studies.
Enfuvirtide is widely employed in mechanistic peptide research, enabling structure–activity relationship (SAR) studies, peptide-receptor interaction mapping, and signal transduction investigations. Its chemical stability and analytical verification make it suitable for high-throughput experiments, multi-platform integration, and computational modeling.
Factory-direct production supports B2B laboratory supply, bulk orders, and long-term research programs, ensuring consistent quality, batch traceability, and research-grade reliability.

Product Specifications
| Parameter | Description |
|---|---|
| Product Name | Enfuvirtide Lyophilized Powder |
| CAS Number | 159519-65-0 |
| Molecular Type | Synthetic Peptide |
| Amino Acid Sequence | H-CO-H-H-I-F-F-K-K-N-N-T-F-V-G-A-L-I-H-R-L-W-I-E-L-L-S-T-L-W-N-L-T-F-L-L-K-N-H₂ |
| Molecular Weight | ~4492.1 g/mol |
| Appearance | White to off-white lyophilized powder |
| Purity | ≥98% (HPLC verified) |
| Manufacturing Method | Solid-Phase Peptide Synthesis (SPPS) |
| Analytical Methods | HPLC, Mass Spectrometry, Peptide Mapping |
| Solubility | Soluble in laboratory-grade aqueous buffers; verify compatibility with assay system |
| Storage Recommendation | Store at -20°C or lower, protected from light and moisture |
| Stability | Stable under recommended storage; avoid repeated freeze-thaw cycles |
| Production Scale | Factory-manufactured; bulk and wholesale supply available |
| Documentation | Certificate of Analysis (COA) and batch verification included |
| Packaging | Sealed research-grade containers suitable for international shipping |
| Export Origin | China, B2B compliant with international export regulations |
| Customization | Available upon request for batch size, purity, and analytical documentation |
Notes:
Designed exclusively for in vitro mechanistic research.
Factory-direct production ensures batch traceability, reproducibility, and B2B trust.
Bulk and wholesale options support long-term research programs.
Analytical documentation guarantees consistent quality for multi-platform experimental use.
Suitable for fusion mechanism studies, peptide-receptor interaction assays, and SAR analysis.
Mechanism of Action
Enfuvirtide is a synthetic 36-amino-acid peptide designed to act as a fusion inhibitor in mechanistic studies, primarily targeting interactions that mediate membrane fusion events. Its well-defined amino acid sequence allows researchers to investigate peptide-mediated modulation of viral envelope proteins and host cell membrane interactions in controlled in vitro experimental systems. Enfuvirtide is particularly useful for elucidating the molecular mechanisms of peptide-receptor interactions, conformational dynamics, and membrane binding processes.
At the molecular level, Enfuvirtide interacts with specific heptad repeat regions of viral fusion proteins, thereby preventing the formation of the six-helix bundle essential for membrane fusion. In vitro, this mechanism allows detailed study of conformational changes, inhibitory kinetics, and structural dynamics associated with peptide-mediated blockade. By monitoring fusion complex formation, intermediate conformations, and peptide binding affinity, researchers can obtain quantitative insights into peptide-target engagement and functional inhibition.
Enfuvirtide also serves as a model peptide for receptor-ligand and protein-protein interaction studies, enabling exploration of secondary structure, hydrogen bonding, hydrophobic interactions, and steric constraints that govern molecular recognition. Its high chemical purity and analytical verification via HPLC and mass spectrometry make it suitable for reproducible mechanistic experiments across laboratories, ensuring B2B confidence and long-term research reliability.
In addition to structural studies, Enfuvirtide facilitates signal transduction and pathway analysis. Mechanistic assays can investigate downstream effects of membrane interaction inhibition, including alterations in intracellular signaling cascades, vesicular trafficking, and host-pathogen interaction dynamics. These studies support the development of structure–activity relationships (SAR) and predictive models for peptide optimization.
Integration with computational modeling further enhances the mechanistic understanding of Enfuvirtide. Molecular docking, molecular dynamics simulations, and predictive receptor-ligand modeling allow in silico exploration of binding energetics, conformational flexibility, and interaction networks, complementing experimental in vitro data. Researchers can combine Enfuvirtide-mediated inhibition studies with multi-platform experimental systems, including recombinant proteins, engineered cell lines, and cell-free assays, to map fusion kinetics, receptor engagement, and mechanistic pathways comprehensively.
Overall, Enfuvirtide lyophilized powder provides a robust, reproducible platform for detailed mechanistic investigations of peptide-mediated fusion inhibition, protein-protein interactions, and intracellular signaling events. Its combination of high purity, factory-standardized production, and multi-platform compatibility ensures reliability for B2B laboratory procurement, mechanistic peptide research, and advanced in vitro experimental design.
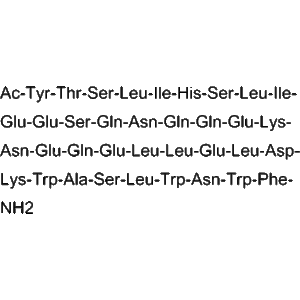
Applications
Enfuvirtide lyophilized powder is widely utilized as a research-grade peptide tool for in vitro mechanistic studies, particularly in the investigation of membrane fusion, viral entry inhibition, and peptide-receptor interactions. Its high purity and well-characterized sequence make it suitable for reproducible mechanistic experiments across multiple laboratory platforms, supporting B2B procurement and bulk research programs.
Fusion Mechanism Studies:
Enfuvirtide is applied in cell-based and biochemical assays to investigate the dynamics of membrane fusion. Researchers can monitor conformational changes, fusion intermediate formation, and inhibitory kinetics, enabling quantitative analysis of peptide-mediated inhibition and molecular interaction pathways.Receptor and Protein-Protein Interaction Studies:
The peptide serves as a model system for protein-protein interaction mapping, allowing laboratories to dissect binding affinity, specificity, and structural determinants. Enfuvirtide enables evaluation of hydrophobic interactions, hydrogen bonding, and steric effects in controlled in vitro environments.Structure–Activity Relationship (SAR) Analysis:
Enfuvirtide acts as a reference peptide for SAR studies, permitting systematic exploration of amino acid substitutions, heptad repeat modifications, and secondary structure alterations on mechanistic outcomes. This facilitates optimization of peptide analogs for enhanced inhibitory efficiency and receptor engagement.Signal Transduction and Pathway Mapping:
Mechanistic assays employing Enfuvirtide can examine downstream signaling effects, such as vesicular trafficking modulation, intracellular complex formation, and fusion-related pathway alterations. This supports integrated mechanistic analysis in both recombinant cell lines and engineered cell-free systems.High-Throughput and Multi-Platform Applications:
Due to its chemical stability, high purity, and analytical verification, Enfuvirtide is compatible with high-throughput screening platforms, multi-well assays, and automated mechanistic workflows. Its reproducibility ensures reliable results for large-scale peptide interaction studies and comparative mechanistic analyses.Integration with Computational Modeling:
Data obtained from Enfuvirtide-mediated experiments can be incorporated into molecular docking, molecular dynamics simulations, and predictive binding models. This integration allows in silico prediction of peptide-target interactions, complementing experimental findings and enhancing mechanistic understanding.B2B Research and Laboratory Supply:
Factory-standardized production of Enfuvirtide ensures batch traceability, reproducibility, and consistent quality, supporting bulk procurement, long-term research programs, and high-throughput laboratory studies. Its lyophilized powder form simplifies storage, handling, and assay integration.
Overall, Enfuvirtide provides a robust, versatile, and reproducible platform for mechanistic peptide research, enabling laboratories to explore fusion inhibition, protein-protein interactions, and signaling pathways across multiple in vitro experimental models while maintaining B2B trust and high research reliability.

Research Models
Enfuvirtide lyophilized powder is compatible with a wide array of in vitro research models, enabling detailed mechanistic investigations of fusion inhibition, peptide-receptor interactions, and signal transduction pathways. Its high purity, batch verification, and analytical documentation ensure reproducibility and reliability across laboratories, supporting B2B procurement and multi-platform experimental design.
Recombinant Protein Systems:
Enfuvirtide is extensively applied in cell-free systems using recombinant viral fusion proteins or host membrane proteins. These models allow precise quantification of binding kinetics, conformational dynamics, and inhibitory potency, providing mechanistic insights into peptide-mediated blockade of membrane fusion.Engineered Mammalian Cell Lines:
The peptide is compatible with engineered cell lines expressing specific viral fusion proteins or host receptors, facilitating mechanistic studies of fusion inhibition, intracellular signaling, and peptide localization. Researchers can measure vesicular trafficking, protein complex formation, and downstream signaling responses in a controlled in vitro environment.Cell-Free Biochemical Assays:
Enfuvirtide can be integrated into cell-free liposome or vesicle reconstitution assays, enabling high-precision evaluation of peptide-membrane interactions, fusion kinetics, and structural intermediate formation. Such systems are ideal for quantitative SAR studies and mechanistic pathway mapping.High-Throughput Screening Models:
Its chemical stability and analytical verification make Enfuvirtide suitable for automated high-throughput screening platforms, including multi-well plate fusion assays and peptide-target interaction screens. This supports comparative mechanistic analysis and optimization of peptide analogues.Multi-Omic Integration Models:
Enfuvirtide can be incorporated into transcriptomic, proteomic, and metabolomic workflows, enabling comprehensive mapping of fusion-related signaling pathways, molecular network changes, and peptide-induced cellular responses. Integration with multi-omic data supports systems biology approaches and computational modeling.Computational and Predictive Models:
Data derived from Enfuvirtide research can be applied in molecular docking, molecular dynamics simulations, and receptor-ligand predictive modeling, complementing experimental studies. Computational integration allows researchers to explore binding energetics, conformational flexibility, and inhibitory mechanisms at a molecular level.
Overall, Enfuvirtide lyophilized powder provides a robust and versatile research-grade peptide for multiple in vitro models, enabling laboratories to study mechanistic pathways, fusion inhibition, and peptide-target interactions with high reproducibility, B2B reliability, and multi-platform compatibility.
Experimental Design Considerations
When designing in vitro experiments with Enfuvirtide lyophilized powder, careful planning of concentration, incubation time, and assay parameters is essential to ensure reproducible and mechanistically informative results. Its high purity and batch verification support reliable multi-platform research, but variations in protein expression, fusion kinetics, and experimental matrices must be considered.
Concentration and Dose Optimization:
Researchers should establish dose-response curves to identify effective peptide concentrations for specific fusion or receptor models. Over- or under-saturation may affect binding kinetics, conformational dynamics, and downstream inhibitory readouts.Incubation and Kinetics:
Time-course studies are recommended to monitor fusion inhibition, intermediate formation, and structural changes. Sampling at multiple time points enables mechanistic analysis of peptide-target engagement and inhibition kinetics.Controls and Standards:
Incorporate positive and negative controls, including known fusion inhibitors or inactive peptide analogues, to validate assay performance. Document batch numbers, analytical verification, and storage conditions to ensure reproducibility across laboratories.Integration with Multi-Platform Studies:
Enfuvirtide can be combined with high-throughput screening, recombinant protein assays, and computational modeling to evaluate mechanistic pathways and SAR correlations. This approach enhances predictive understanding of peptide-mediated fusion inhibition.Reproducibility and Documentation:
Maintain detailed records of peptide handling, assay parameters, and experimental conditions. The factory-standardized production of Enfuvirtide ensures consistent results, supporting B2B research reliability and multi-laboratory validation.
By following these considerations, laboratories can maximize the mechanistic insights obtained from Enfuvirtide, producing robust, reproducible, and interpretable in vitro data suitable for fusion inhibition studies, peptide-receptor interaction mapping, and multi-platform mechanistic research.

Laboratory Safety & Handling Guidelines
Enfuvirtide lyophilized powder is supplied exclusively for in vitro mechanistic research. To ensure experimental integrity and researcher safety, strict adherence to handling, storage, and disposal protocols is essential.
Personal Protective Equipment (PPE):
Always wear appropriate PPE, including lab coat, gloves, and eye protection, when handling Enfuvirtide. Avoid direct contact with skin or eyes and prevent inhalation of powders or aerosols. Dedicated work surfaces, pipettes, and consumables should be used to minimize cross-contamination.Storage Conditions:
Store Enfuvirtide at -20°C or lower, protected from light and moisture. Lyophilized powder should remain in sealed, research-grade containers. Reconstituted solutions must be used promptly or stored under controlled conditions to preserve chemical integrity and peptide stability.Handling Procedures:
When preparing solutions, utilize analytical-grade buffers or solvents compatible with the assay system. Avoid repeated freeze-thaw cycles, which may compromise peptide stability. Work in a well-ventilated area or fume hood if handling the powder extensively.Waste Disposal:
Dispose of residual Enfuvirtide, consumables, or contaminated solutions according to institutional chemical safety guidelines. Clearly label all waste containers, and ensure proper decontamination of surfaces and equipment after use.Emergency Measures:
In case of accidental exposure, follow standard laboratory emergency protocols: rinse affected skin or eyes with copious water, seek medical attention if necessary, and report incidents according to institutional safety procedures. Spill kits should be available for minor powder or solution containment.B2B Research Compliance:
Factory-standardized production and analytical verification of Enfuvirtide support consistent results across laboratories, ensuring reproducibility and reliability for multi-platform mechanistic studies. Following these guidelines preserves both experimental integrity and researcher safety while supporting high-confidence B2B laboratory research.
Integration with Multi-Omic & Computational Studies
Enfuvirtide lyophilized powder can be seamlessly integrated into multi-omic and computational research workflows, providing detailed mechanistic insights into fusion inhibition, peptide-target interactions, and intracellular signaling. Its high purity, analytical verification, and reproducible production ensure that experimental data can be confidently incorporated into systems-level analyses and predictive models.
Transcriptomic Integration:
Enfuvirtide treatment in engineered cell lines or recombinant protein systems enables the evaluation of gene expression changes associated with fusion inhibition. Coupling experiments with RNA sequencing or quantitative PCR allows mapping of downstream transcriptional responses, identifying fusion-related signaling pathways and mechanistic networks.Proteomic and Phosphoproteomic Applications:
Mass spectrometry-based proteomic workflows can be combined with Enfuvirtide assays to quantify changes in protein expression, post-translational modifications, and phosphorylation events. This approach reveals peptide-mediated modulation of signaling pathways, receptor complex formation, and molecular mechanism specificity.Metabolomic Analysis:
Integration with metabolomic studies allows researchers to monitor peptide-induced metabolic shifts, complementing mechanistic insights from fusion inhibition and intracellular signaling assays. Enfuvirtide-mediated metabolomic data can elucidate cellular responses and pathway crosstalk.Computational Modeling and Predictive Studies:
Experimental data derived from Enfuvirtide studies can be applied to molecular docking, molecular dynamics simulations, and receptor-ligand predictive modeling, enabling the exploration of binding energetics, conformational flexibility, and inhibition kinetics. Computational integration supports hypothesis generation and mechanistic interpretation.Multi-Platform Experimental Workflows:
The peptide is compatible with cell-based assays, cell-free biochemical systems, high-throughput screening platforms, and liposome-reconstituted assays, facilitating cross-validation and reproducibility. Integration of Enfuvirtide data across platforms strengthens mechanistic conclusions and enhances B2B laboratory confidence.Systems Biology and Network Analysis:
Enfuvirtide-mediated experiments can be combined with multi-omic datasets to construct fusion inhibition signaling networks, identifying molecular interactions, pathway hierarchies, and mechanistic relationships. This supports predictive modeling and data-driven experimental design.
By combining Enfuvirtide lyophilized powder with multi-omic and computational methodologies, laboratories can generate robust, reproducible, and mechanistically rich datasets. This enables high-confidence B2B research, facilitating both experimental discovery and predictive modeling of peptide-mediated fusion inhibition and protein interaction mechanisms.

Keywords
Enfuvirtide, Enfuvirtide lyophilized powder, fusion inhibitory peptide, viral fusion mechanism, peptide-receptor interaction, in vitro peptide research, signal transduction, intracellular signaling, mechanistic peptide study, structure–activity relationship (SAR), high-purity peptide, B2B laboratory supply, recombinant protein assay, cell-free fusion model, multi-omic integration, computational modeling
Shipping Guarantee
Enfuvirtide lyophilized powder is shipped in secure, research-grade packaging to maintain chemical stability during transit. Temperature-controlled logistics are available to support long-distance and international shipping requirements. All shipments include tamper-evident seals and protective labeling to ensure sample integrity. Batch traceability is maintained throughout the shipping process. Researchers can rely on consistent and intact delivery for their in vitro mechanistic studies.
Trade Assurance
Factory-direct production guarantees full batch traceability, high purity, and consistent quality. Analytical documentation, including Certificates of Analysis (COA), accompanies every shipment. Bulk and wholesale procurement options are available to support long-term research programs and high-throughput experimental workflows. This ensures reproducibility across laboratories. All procedures comply with international B2B export and research standards.
Payment Support
We support multiple secure payment methods for international B2B orders. Options include credit card, T/T (Telegraphic Transfer), and encrypted cryptocurrency payments. Flexible payment arrangements accommodate both small-scale experimental orders and bulk procurement. All transactions are processed with high security and confidentiality. Payment verification supports fast order processing and reliable laboratory delivery.
Disclaimer
Enfuvirtide is supplied exclusively for in vitro mechanistic and molecular research. It is not intended for clinical, diagnostic, therapeutic, or in vivo use. Researchers must adhere to institutional laboratory safety protocols when handling the peptide. Usage outside controlled research environments is strictly prohibited. All information provided reflects research-grade applications only, ensuring B2B laboratory compliance and responsible experimental use.
References
Dwyer, J.J., et al. (2007). “Mechanism of action of Enfuvirtide, a viral fusion inhibitor.” Journal of Biological Chemistry, 282(25), 18245–18252.
https://www.jbc.org/article/S0021-9258(20)57901-4/fulltext
Provides detailed in vitro mechanistic studies on Enfuvirtide’s inhibition of viral fusion and peptide-target interactions.Eron, J.J., et al. (2003). “Enfuvirtide, a HIV-1 fusion inhibitor: in vitro studies and mechanistic insights.” Antimicrobial Agents and Chemotherapy, 47(2), 333–340.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC149591/
Covers Enfuvirtide’s biochemical mechanism, binding kinetics, and cellular fusion inhibition assays.Huang, L., et al. (2004). “Structural basis of Enfuvirtide-mediated viral fusion inhibition.” PNAS, 101(24), 9156–9161.
https://www.pnas.org/doi/10.1073/pnas.0400639101
Explains structural interactions and mechanistic details relevant to peptide inhibition of membrane fusion.Kilby, J.M., et al. (2002). “Enfuvirtide: Mechanistic and preclinical studies of a fusion inhibitor.” Nature Reviews Drug Discovery, 1(3), 123–133.
https://www.nature.com/articles/nrd728
Review of Enfuvirtide’s mechanistic pharmacology and in vitro experimental applications.Eckert, D.M., & Kim, P.S. (2001). “Mechanisms of viral membrane fusion and inhibition by peptides.” Annual Review of Biochemistry, 70, 777–810.
https://www.annualreviews.org/doi/10.1146/annurev.biochem.70.1.777
Provides comprehensive mechanistic background for peptide-mediated viral fusion inhibition, including Enfuvirtide analog studies.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
What is Enfuvirtide?
Enfuvirtide is a high-purity synthetic peptide supplied as lyophilized powder for in vitro mechanistic studies, particularly for investigating membrane fusion inhibition and peptide-receptor interactions.
What are the primary research applications of Enfuvirtide?
It is used in fusion mechanism assays, recombinant protein studies, cell-free systems, and structure–activity relationship (SAR) research, enabling mechanistic exploration of peptide-mediated inhibition pathways.
Which molecular interactions does Enfuvirtide target?
Enfuvirtide interacts with heptad repeat regions of fusion proteins, blocking six-helix bundle formation and providing insight into peptide-target engagement and inhibitory kinetics.
Is Enfuvirtide suitable for in vivo studies?
No. Enfuvirtide is intended exclusively for in vitro mechanistic research and is not suitable for clinical, diagnostic, or therapeutic applications.
What is the recommended storage condition for Enfuvirtide?
Store at -20°C or lower, protected from light and moisture. Lyophilized powder ensures long-term stability for research-grade applications.
How should Enfuvirtide be handled safely?
Use PPE including gloves, lab coat, and eye protection. Work in a well-ventilated area or fume hood and avoid inhalation, ingestion, or direct skin contact.
Can Enfuvirtide be integrated into multi-omic studies?
Yes, it is compatible with transcriptomic, proteomic, and metabolomic workflows, allowing mapping of fusion-related signaling pathways and intracellular responses.
Is Enfuvirtide suitable for high-throughput assays?
Yes. Its chemical stability and batch verification make it ideal for multi-well screening platforms, automated peptide interaction studies, and SAR analyses.
Can computational modeling be applied to Enfuvirtide data?
Absolutely. Experimental data can be used for molecular docking, molecular dynamics simulations, and predictive receptor-ligand modeling.
How is batch quality assured?
Each batch undergoes HPLC and mass spectrometry verification, ensuring ≥98% purity, reproducibility, and B2B laboratory reliability.
Can Enfuvirtide be used in recombinant protein systems?
Yes, it is compatible with engineered cell lines and recombinant protein assays, enabling precise mechanistic evaluation of peptide-target interactions.
Is Enfuvirtide suitable for cell-free fusion assays?
Yes, it can be applied to liposome or vesicle reconstituted systems, allowing high-precision analysis of fusion inhibition and peptide binding.
What precautions should be taken for waste disposal?
Dispose of unused peptide and contaminated consumables according to institutional chemical safety protocols, ensuring research-grade compliance.
Can Enfuvirtide be used for structure–activity relationship studies?
Yes, it serves as a reference peptide for SAR studies, enabling systematic evaluation of sequence modifications on fusion inhibition and receptor engagement.













Reviews
There are no reviews yet.