No products in the cart.
Dynorphin A – Purity 98.18%
$2.00
Dynorphin A is a high-purity endogenous peptide supplied as a lyophilized powder, manufactured under controlled factory conditions for in vitro mechanistic research. It is widely used as a molecular probe in receptor–ligand interaction studies, peptide signaling analysis, and biochemical pathway investigations. Bulk and wholesale supply is available for global research institutions and industrial laboratories.
Description
Contents
hide
Product Description
Dynorphin A is a naturally occurring opioid-derived peptide composed of a defined amino acid sequence that has become an essential molecular tool in modern biochemical and receptor signaling research. Supplied as a high-purity lyophilized powder, Dynorphin A is specifically designed for controlled laboratory use, enabling reproducible experimentation in in vitro and molecular-level studies.
From a structural perspective, Dynorphin A exhibits well-characterized peptide conformation and charge distribution, making it suitable for detailed investigation of peptide–receptor binding behavior, affinity modulation, and competitive interaction analysis. Its molecular integrity and sequence fidelity are verified through HPLC and mass spectrometry, ensuring consistency across experimental batches.
In research environments, Dynorphin A is commonly employed as a reference peptide to explore receptor selectivity, ligand specificity, and downstream signal transduction processes. The peptide’s stability in lyophilized form allows researchers to precisely control reconstitution parameters, concentration gradients, and experimental exposure conditions, all of which are critical for quantitative mechanistic studies.
The product is manufactured in a certified peptide synthesis facility in China, following standardized production and purification workflows. This ensures batch-to-batch reproducibility, traceability, and scalability, making Dynorphin A suitable for both small-scale academic studies and large-volume industrial R&D projects. Factory-direct supply enables cost-efficient bulk procurement, supporting long-term research programs and high-throughput assay development.
Importantly, Dynorphin A offered here is positioned strictly as a research-grade material. All technical documentation, analytical reports, and handling recommendations are aligned with laboratory research standards, ensuring that investigators can confidently integrate this peptide into advanced biochemical assays, receptor interaction studies, and molecular signaling research pipelines.
Product Specifications
| Specification Category | Details |
|---|---|
| Product Name | Dynorphin A |
| Synonyms | Dynorphin A peptide |
| Molecular Type | Synthetic linear peptide |
| Amino Acid Length | Defined endogenous peptide sequence |
| Molecular Structure | Precisely characterized peptide backbone |
| Form | Lyophilized powder |
| Appearance | White to off-white amorphous solid |
| Purity | ≥98% / ≥99% (HPLC) |
| Identity Confirmation | Mass Spectrometry (MS) |
| Counter Ion | None (or specified upon customization) |
| Solubility | Suitable for controlled laboratory reconstitution |
| Packaging | Sterile glass vials (customizable sizes) |
| Storage Conditions | Sealed, low-temperature, light-protected environment |
| Stability | Stable under recommended laboratory storage conditions |
| Intended Application | In vitro laboratory research only |
| Manufacturing Standard | Factory-manufactured under controlled conditions |
| Supply Mode | Bulk, wholesale, and customized B2B supply |
| Country of Origin | China (global export supported) |
Specification Notes & Research Relevance
Dynorphin A is supplied as a research-grade lyophilized peptide, enabling precise control over reconstitution concentration, experimental exposure, and assay reproducibility. The lyophilized format minimizes degradation risk during transportation and long-term storage, making it suitable for extended research programs and high-throughput workflows.
Purity levels of ≥98% or ≥99% are achieved through advanced purification processes and verified using high-performance liquid chromatography (HPLC), while mass spectrometry (MS) confirms molecular identity and sequence integrity. These analytical validations ensure consistency across batches and reliability in quantitative mechanistic experiments.
The absence of unnecessary excipients or fillers allows Dynorphin A to be directly integrated into biochemical assays, receptor-binding studies, and peptide interaction analyses without interference. Custom vial sizes and batch quantities are available to accommodate both small-scale exploratory research and large-volume industrial R&D needs.
Manufactured in a certified peptide synthesis facility, each batch is fully traceable and supported by quality control documentation, including analytical reports and production records. Factory-direct production ensures cost-efficient pricing, stable supply, and scalability, making Dynorphin A an ideal choice for laboratories seeking high-purity peptide materials with reliable wholesale availability.
Mechanism of Action
Dynorphin A is a well-characterized endogenous peptide widely utilized in in vitro molecular mechanism studies to investigate peptide–receptor interactions and downstream signaling processes. Its defined amino acid sequence and stable structural properties allow researchers to precisely examine ligand binding behavior, receptor selectivity, and signal initiation events within controlled laboratory environments.
At the molecular level, Dynorphin A functions as a high-affinity peptide ligand, enabling detailed analysis of receptor engagement and conformational modulation. In receptor-expressing systems and purified protein assays, Dynorphin A supports the investigation of binding kinetics, competitive displacement, and affinity ranking when compared with structurally related peptides or synthetic analogs. These studies are essential for understanding structure–function relationships in peptide signaling research.
Dynorphin A is frequently applied in cell-free and receptor-isolated platforms, where its interaction with target receptors can be monitored without confounding systemic variables. This allows for high-resolution measurement of molecular signaling initiation, receptor activation states, and downstream biochemical responses. The peptide’s consistent purity ensures that observed effects can be confidently attributed to specific molecular interactions rather than batch variability.
In mechanistic research workflows, Dynorphin A is also used to explore signal modulation pathways, including receptor-associated second-messenger systems and protein–protein interaction networks. By combining experimental data with quantitative analytical techniques, researchers can map signaling cascades and identify critical nodes of regulation.
Furthermore, Dynorphin A serves as a valuable reference compound in structure–activity relationship (SAR) and comparative peptide studies. Integration with computational modeling, such as molecular docking and dynamic simulations, enables correlation between experimental binding data and predicted interaction models, providing a comprehensive understanding of peptide-mediated molecular mechanisms.
Overall, Dynorphin A functions as a robust molecular probe for elucidating receptor-level mechanisms, peptide signaling dynamics, and structural determinants of ligand activity in advanced in vitro research settings.
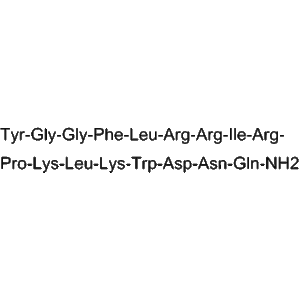
Applications
Dynorphin A is extensively utilized across advanced laboratory research disciplines that focus on peptide signaling, receptor interaction analysis, and molecular mechanism elucidation. As a high-purity peptide supplied in lyophilized form, it provides researchers with a reliable and reproducible molecular tool for controlled in vitro experimental systems.
One primary application of Dynorphin A is in receptor–ligand binding studies, where it is used to characterize binding affinity, receptor selectivity, and competitive interaction dynamics. In purified receptor preparations or engineered receptor-expression platforms, Dynorphin A serves as a benchmark peptide for comparing interaction profiles with synthetic analogs, truncated sequences, or modified ligands. These studies are fundamental to understanding peptide recognition at the molecular level.
Dynorphin A is also widely applied in biochemical signaling pathway research. In cell-free or isolated signaling systems, researchers employ the peptide to investigate signal initiation events, receptor-associated conformational changes, and downstream molecular responses. Such experiments contribute to mapping peptide-driven signaling networks and identifying key regulatory nodes within these pathways.
In structure–activity relationship (SAR) investigations, Dynorphin A enables systematic evaluation of sequence-dependent functional properties. By comparing the native peptide with sequence variants, researchers can determine which residues or motifs are critical for receptor engagement and signaling modulation. This application is particularly valuable for peptide engineering and rational design studies conducted in controlled laboratory environments.
High-throughput research platforms further benefit from Dynorphin A as a standardized reference compound. Its verified purity and batch-to-batch consistency support assay validation, quality control, and comparative screening workflows. This makes it suitable for large-scale experimental programs within academic laboratories, CROs, and industrial R&D facilities.
Additionally, Dynorphin A plays a role in integrative computational and data-driven research. Experimental findings generated using this peptide can be combined with molecular docking, dynamic simulations, and network modeling to enhance mechanistic interpretation. This integrative approach supports hypothesis testing and predictive modeling of peptide–receptor interactions.
Overall, Dynorphin A is a versatile and essential research reagent, enabling precise mechanistic exploration, comparative analysis, and reproducible peptide research across a wide range of in vitro laboratory applications.
Research Models
Dynorphin A is compatible with a wide range of in vitro research models designed to investigate peptide–receptor interactions, molecular signaling mechanisms, and structural determinants of ligand activity. These models provide controlled experimental environments that allow precise manipulation of variables and high reproducibility across studies.
One commonly used model involves cell-free biochemical systems, where purified receptors, binding proteins, or signaling components are isolated from complex biological matrices. In such systems, Dynorphin A can be applied to assess binding affinity, kinetic parameters, and competitive displacement behavior with minimal background interference. This approach is particularly valuable for quantitative analysis and mechanistic validation.
Recombinant receptor-expression platforms represent another important research model. Using engineered systems that express defined receptor subtypes, researchers can explore ligand selectivity and receptor-specific interaction profiles. Dynorphin A serves as a reference peptide in these models, enabling comparative studies across receptor variants and facilitating detailed mapping of receptor–ligand recognition patterns.
Dynorphin A is also employed in high-throughput screening (HTS)–compatible models, where standardized assay formats require consistent reagents with verified purity. Its stability and batch-to-batch consistency support parallel testing of multiple peptides or compounds, ensuring reliable comparative outcomes. These models are widely used in industrial research and contract research organizations.
For structural and mechanistic exploration, biophysical research models such as surface interaction platforms and conformational analysis systems allow researchers to study molecular binding events and structural transitions induced by peptide interaction. Data generated from these models contribute to a deeper understanding of peptide-driven molecular mechanisms.
Finally, Dynorphin A is increasingly integrated into hybrid experimental–computational models, where in vitro findings are combined with simulation and modeling approaches. This synergy enhances predictive accuracy and supports comprehensive mechanistic interpretation.
Overall, these research models establish Dynorphin A as a versatile and reliable peptide tool for advanced laboratory investigations into receptor biology and peptide-mediated molecular processes.
Experimental Design Considerations
Effective use of Dynorphin A in laboratory research requires well-structured experimental design to ensure data reliability, reproducibility, and mechanistic clarity. Prior to full-scale experimentation, researchers are encouraged to establish baseline control systems that exclude peptide exposure, allowing differentiation between specific peptide-driven effects and background assay responses.
Initial concentration-range optimization is recommended through pilot studies. Gradual titration enables identification of interaction windows suitable for quantitative analysis while minimizing non-specific binding or signal saturation. Consistent reconstitution procedures, including solvent selection and mixing conditions, should be standardized and documented to maintain experimental consistency across batches.
Temporal parameters represent another critical design element. Clearly defined incubation times, sampling intervals, and endpoint measurements help capture dynamic peptide–receptor interactions and downstream molecular responses. Synchronization of timing across experimental replicates supports meaningful comparative analysis.
Selection of appropriate assay formats and analytical readouts should align with the specific mechanistic questions being investigated, whether focusing on binding kinetics, competitive displacement, or signaling pathway modulation. Inclusion of reference peptides or structural analogs further enhances interpretability and supports structure–activity relationship analysis.
Comprehensive documentation of batch identifiers, storage conditions, and experimental variables is essential, particularly for collaborative or multi-site research projects. By adhering to these design considerations, Dynorphin A can be employed as a robust and reproducible molecular tool in advanced in vitro mechanistic studies.
Laboratory Safety & Handling Guidelines
Dynorphin A is supplied as a research-grade lyophilized peptide and must be handled exclusively within professional laboratory environments that follow established safety and quality management protocols. Although intended solely for in vitro research, appropriate laboratory practices are essential to maintain material integrity and ensure a safe working environment.
All handling procedures should be conducted by trained personnel wearing standard personal protective equipment (PPE), including laboratory coats, protective gloves, and safety eyewear. Handling should take place on clean, designated laboratory benches to minimize contamination and ensure experimental reproducibility. Direct contact with the material should be avoided, and tools used during handling should be sterile and appropriate for peptide manipulation.
Prior to use, Dynorphin A should remain in its original, sealed container under recommended storage conditions. Reconstitution, when required, must be performed using validated laboratory-grade solvents and controlled techniques. Gentle mixing is advised to preserve peptide integrity and prevent structural degradation. All reconstitution parameters should be clearly documented for reproducibility.
Storage conditions play a critical role in maintaining peptide stability. Dynorphin A should be stored in a low-temperature, light-protected environment, with containers tightly sealed to prevent moisture exposure. Repeated temperature fluctuations and unnecessary container opening should be avoided to reduce the risk of degradation.
Work areas should be clearly labeled, and cross-contamination with other research materials must be prevented through dedicated tools and consumables. After experimental use, residual material, disposable consumables, and contaminated items should be disposed of in accordance with institutional chemical waste management guidelines and local regulatory requirements.
Routine inspection of storage containers, labeling, and documentation is recommended to ensure continued compliance with laboratory standards. By adhering to these laboratory safety and handling guidelines, researchers can ensure consistent experimental outcomes, material integrity, and safe laboratory operations when working with Dynorphin A in advanced in vitro research settings.
Integration with Multi-Omic & Computational Studies
Dynorphin A is well suited for integration into multi-omic research frameworks and computational modeling workflows, enabling comprehensive analysis of peptide-mediated molecular mechanisms at multiple analytical levels. When applied in controlled in vitro systems, data generated using Dynorphin A can be effectively combined with proteomic, transcriptomic, and interactomic datasets to provide a broader understanding of receptor-associated signaling processes.
In proteomic studies, Dynorphin A supports the identification of receptor-associated protein complexes, downstream signaling partners, and post-interaction molecular changes. Quantitative proteomic outputs can be correlated with peptide–receptor binding data to map signaling cascades and regulatory nodes. This approach enhances mechanistic resolution beyond single-pathway analysis.
Transcriptomic integration allows researchers to explore gene expression patterns linked to receptor engagement and signal modulation in experimental systems. By aligning transcript-level data with biochemical assay results, investigators can establish associations between peptide-driven receptor interactions and broader molecular response networks.
Computational methodologies further strengthen these analyses. Molecular docking and dynamic simulations enable prediction and visualization of peptide–receptor interaction modes, supporting interpretation of experimental binding data. These in silico approaches help identify critical interaction residues and conformational changes that may not be directly observable through experimental assays alone.
Network-based modeling and pathway analysis tools allow researchers to integrate multi-omic datasets with experimental observations, facilitating systems-level interpretation and hypothesis generation. By iteratively refining computational models using empirical data derived from Dynorphin A studies, researchers can improve predictive accuracy and experimental design efficiency.
Overall, the integration of Dynorphin A into multi-omic and computational research pipelines supports holistic, data-driven exploration of peptide signaling mechanisms, enabling deeper insight into molecular interactions and advancing high-level mechanistic research in laboratory settings.
Keywords
Dynorphin A, Dynorphin A peptide, Dynorphin A lyophilized powder, high purity Dynorphin A, peptide receptor research, in vitro peptide studies, factory manufactured peptide, wholesale peptide supplier, China B2B peptide export
Shipping Guarantee
Dynorphin A is shipped using research-grade protective packaging designed to preserve peptide integrity throughout international transit. Packaging solutions minimize exposure to moisture, light, and physical stress, ensuring consistent material quality upon arrival. Export logistics are optimized for global laboratory delivery, with standardized documentation supporting smooth customs clearance. Each shipment is handled in accordance with international laboratory supply standards to maintain product stability. This shipping framework supports reliable delivery for both small-volume orders and bulk wholesale consignments.
Trade Assurance
All Dynorphin A products are supplied through factory-direct manufacturing channels, ensuring full batch traceability and quality consistency. Each batch is supported by analytical documentation, including purity verification and identity confirmation. Long-term cooperation models are available for CROs, research institutions, and industrial R&D partners, supporting stable supply and scalable procurement. Wholesale and bulk purchasing options are designed to meet ongoing research demands with cost efficiency. Dedicated export processes reinforce trust and transparency in international B2B transactions.
Payment Support
Flexible payment options are provided to support global B2B research procurement workflows. Accepted methods include Credit Card, T/T (Telegraphic Transfer), and Cryptocurrency with encrypted payment channels. Payment processes are designed to be secure, efficient, and compliant with international trade practices. Customized payment arrangements are available for long-term partners and bulk purchasers. This multi-option payment structure ensures accessibility for research organizations worldwide.
Disclaimer
This product is strictly intended for laboratory research use only.
It is not for diagnostic, therapeutic, clinical, or any non-research applications.
All information provided is for scientific, educational, and experimental reference purposes within controlled laboratory environments.
The supplier assumes no responsibility for use outside the scope of qualified laboratory research.
References
Dynorphin A sequence and receptor interaction background – R&D Systems
Peptide catalog entry with sequence and high‑purity context for laboratory receptor research. www.rndsystems.comAnalysis of Dynorphin and Dynorphin Receptor Interactions
Peer‑reviewed article detailing structure–activity relationships and receptor binding insights.https://go.drugbank.com/articles/A29528Dynorphin peptides differential regulation at kappa receptors – PubMed
Research study on peptide–receptor regulation and interaction characteristics. PubMedStructure and dynamics of dynorphin peptides and receptors – PubMed
Review on peptide structure and receptor binding dynamics in molecular research. PubMedPorcine pituitary dynorphin structural characterization – PMC
Foundational peptide sequencing and structural data for Dynorphin A. PMC
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
1. What is Dynorphin A used for in research?
Dynorphin A is primarily used as a molecular probe in in vitro receptor–ligand studies. It helps researchers investigate binding kinetics, receptor selectivity, and downstream signaling mechanisms. All applications are restricted to controlled laboratory environments.
2. What purity level is provided?
This product is supplied at ≥98% or ≥99% purity, confirmed via HPLC and mass spectrometry. High purity ensures reproducibility and reliability in quantitative mechanistic studies. Analytical documentation is included for batch verification.
3. What form does Dynorphin A come in?
Dynorphin A is provided as a lyophilized powder, facilitating controlled reconstitution and precise experimental dosing. Lyophilization ensures stability during storage and shipment. Reconstitution instructions should be followed for optimal results.
4. Can Dynorphin A be used for in vivo studies?
No. Dynorphin A is strictly for in vitro laboratory research. It is not intended for clinical, therapeutic, animal, or human applications. Use should be limited to controlled experimental systems.
5. How should Dynorphin A be stored?
Store in a sealed container at 2–8 °C in a light-protected environment. Avoid repeated freeze-thaw cycles and prolonged exposure to ambient conditions. Proper storage maintains peptide integrity and reproducibility.
6. Is batch traceability provided?
Yes. Each batch includes quality control documentation, analytical certificates, and production records. This ensures reliability and reproducibility for multi-site or collaborative research projects.
7. Can Dynorphin A be integrated with computational studies?
Yes. Experimental data can be combined with molecular docking, dynamic simulations, and receptor modeling. This integration enables comprehensive mechanistic interpretation and predictive research insights.
8. What types of assays are suitable?
Dynorphin A is compatible with binding assays, receptor-expressing systems, high-throughput screening, and SAR studies. Standardized protocols are recommended to ensure consistent results.
9. How should residual material be disposed of?
Waste should be handled according to institutional chemical waste protocols. All contaminated consumables must be disposed of safely. Following guidelines ensures laboratory safety and regulatory compliance.
10. Can Dynorphin A be shipped internationally?
Yes. It is supplied for global B2B export from China. Shipments use temperature-controlled packaging and include full documentation to preserve integrity and support customs clearance.
11. Are recommended controls necessary?
Yes. Include baseline controls and structurally related peptide comparisons to confirm specificity of observed effects. Controls are essential for accurate mechanistic conclusions.
12. Can Dynorphin A be used in multi-omic studies?
Yes. It can integrate with proteomics, transcriptomics, and interactomics, allowing correlation of receptor interactions with broader molecular pathways. This supports comprehensive mechanistic research.
13. What precautions should be taken during handling?
Use personal protective equipment (PPE), including gloves, lab coat, and eyewear. Work on designated clean surfaces and follow all laboratory safety protocols. This ensures experimental integrity and personnel safety.
14. Is Dynorphin A compatible with automated systems?
Yes. Its lyophilized format supports automated liquid handling and high-throughput experimental workflows. This ensures precise dosing and reproducible results across multiple assays.
15. Can Dynorphin A be used for SAR or comparative studies?
Yes. It serves as a reference peptide for evaluating structure–activity relationships. Researchers can compare full-length sequences, truncated analogs, or modified peptides to elucidate residue-specific effects on receptor binding and signaling.

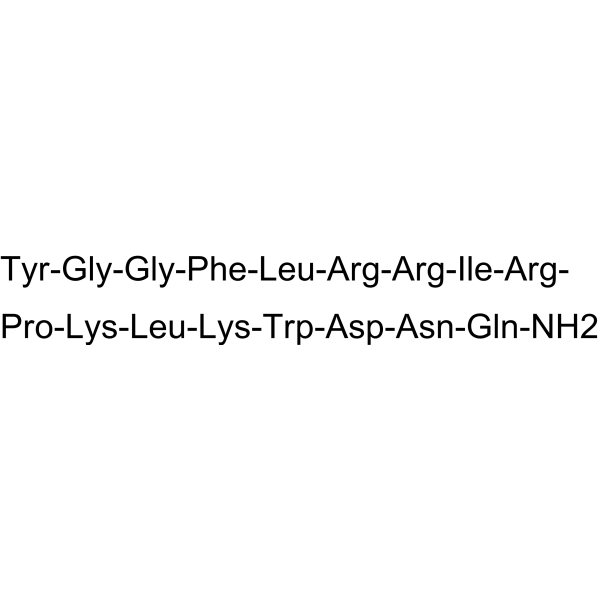







Reviews
There are no reviews yet.