No products in the cart.
GNQWFI – GMP Manufacturer
$2.00
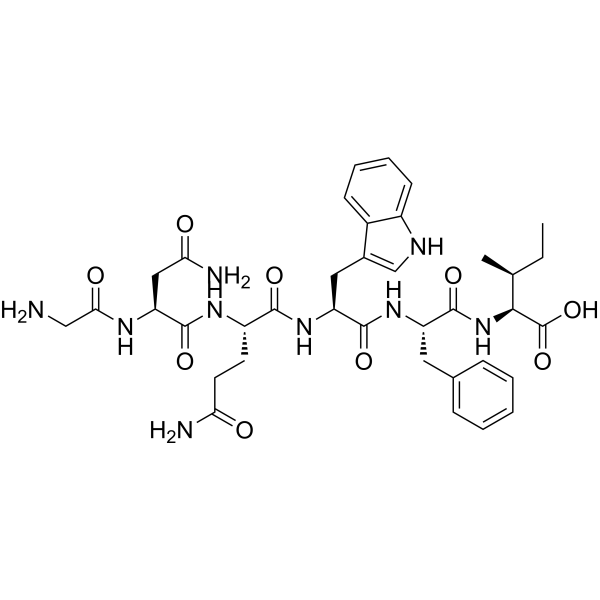
GNQWFI is a GMP-grade anti-Flt1 hexapeptide (VEGFR1-specific antagonist) with 99.45% purity. It blocks VEGFR1 interactions with VEGFA/VEGFB/PlGF and inhibits VEGF-induced endothelial migration and tube formation. Available for wholesale and retail. For laboratory research use only.?For wholesale prices, other specifications and uses, please consult our staff?
Description
GNQWFI is a rationally designed anti-Flt1 hexapeptide that functions as a VEGFR1-specific antagonist. By occupying ligand-contact interfaces on the VEGFR1 (Flt-1) ectodomain, GNQWFI prevents productive binding of VEGFA, VEGFB, and placental growth factor (PlGF)—three principal VEGFR1 ligands implicated in angiogenic and inflammatory signaling. Functionally, this selective blockade suppresses VEGF-induced endothelial cell migration and capillary-like tube formation in vitro, making GNQWFI a practical tool compound for dissecting receptor-specific roles within the VEGF axis.
VEGFR1 is increasingly recognized as a regulatory checkpoint in pathological angiogenesis, vascular permeability, leukocyte recruitment, and tissue remodeling. In oncology, decoupling VEGFR1-mediated cues can reduce pro-angiogenic support within the tumor microenvironment and may influence metastatic conditioning in preclinical models. In ocular disease research, selective interference with VEGFR1 signaling is of interest for retinal and choroidal neovascular pathologies, where permeability and aberrant vessel growth drive vision loss. In airway biology and asthma, abnormal angiogenesis and vascular remodeling correlate with airway hyperreactivity; receptor-focused antagonists like GNQWFI allow controlled evaluation of VEGFR1’s contribution to these processes.
This product is produced under GMP-compliant manufacturing with 99.45% HPLC purity, supporting reproducible results across labs. It is supplied as a stable, lyophilized powder suitable for aqueous or mixed-solvent stock preparation, with standard research pack sizes for screening and scale-appropriate bulk options for extended studies or method development. Each lot is shipped with a comprehensive quality dossier (typical: CoA, HPLC trace, MS identity) to streamline method transfer and documentation in regulated or GLP-adjacent settings.
Product Specifications
| Parameter | Details |
|---|---|
| Product Name | GNQWFI |
| Synonyms | Anti-Flt1 peptide; VEGFR1 antagonist hexapeptide; anti-VEGFR1 peptide |
| CAS No. | 853995-60-5 |
| Identity | Hexapeptide antagonist of VEGFR1 (Flt-1) |
| Sequence (one-letter) | G-N-Q-W-F-I (Gly-Asn-Gln-Trp-Phe-Ile) |
| Target | VEGFR1 (Flt-1) – ligand binding interface |
| Purity | 99.45% (HPLC, GMP-grade) |
| Form | Lyophilized powder |
| Appearance | White to off-white solid |
| Solubility | Readily soluble in water, PBS, and DMSO; prepare sterile-filtered working stocks as required |
| Stability/Storage | Store at –20 °C (desiccated, light-protected). For long-term storage, keep sealed under inert conditions if possible. Avoid repeated freeze–thaw. |
| Reconstitution | Reconstitute in sterile water or buffer; for sensitive assays, include carrier protein or appropriate excipient per lab SOPs |
| Endotoxin | Research grade; typical ? 1.0 EU/mg (lot-specific; see CoA) |
| Manufacturing Standard | GMP-compliant synthesis and purification |
| Applications | Angiogenesis/vascular biology research; tumor microenvironment studies; ocular neovascular models; asthma/airway remodeling models |
| Regulatory Status | For laboratory research use only. Not for human or veterinary use. |
Mechanism of Action & Research Applications
Mechanism of Action
GNQWFI exerts competitive antagonism at VEGFR1. The peptide’s hexameric motif engages receptor surfaces that normally accommodate VEGFA, VEGFB, and PlGF, thereby blocking upstream ligand–receptor engagement. Without productive ligand docking, downstream pathways (e.g., PI3K/AKT, FAK/Src, and small GTPase programs associated with motility and cytoskeletal remodeling) are attenuated, which translates to reduced endothelial chemotaxis, suppressed sprouting, and impaired tube formation in standardized angiogenesis assays. Because GNQWFI is VEGFR1-focused, it enables experiments that isolate Flt-1 contributions from VEGFR2/KDR-dominant signaling, a longstanding technical need in angiogenesis biology.
Research Applications (Illustrative Use Cases)
Oncology and Metastasis Biology
Probe how VEGFR1 blockade influences tumor-associated macrophage recruitment, pre-metastatic niche conditioning, or stromal vascular support.
Combine GNQWFI with VEGFR2 inhibitors or anti-integrin reagents to map pathway redundancy and identify compensatory pro-angiogenic circuits.
Ocular Neovascular Diseases
Evaluate effects on choroidal/retinal neovascularization, vascular leakage, and edema in preclinical models.
Explore combination strategies with agents targeting Tie2/Ang pathways or complement cascades to test multi-node anti-leakage hypotheses.
Asthma and Airway Remodeling
Examine the role of VEGFR1-dependent vascular remodeling in airway hyperresponsiveness using GNQWFI to alter peribronchial angiogenesis.
Quantify changes in microvessel density, vascular permeability, and inflammatory cell trafficking in challenge models.
Fundamental Angiogenesis & Vascular Permeability
Deploy GNQWFI in endothelial scratch/wound assays, transwell migration, and Matrigel tube formation to benchmark VEGFR1-specific contributions.
Integrate with phospho-proteomics or single-cell RNA-seq to deconvolute receptor-biased signaling at pathway resolution.
Biomaterials & Targeted Delivery
Incorporate GNQWFI into nanoparticle or hydrogel systems to assess localized VEGFR1 antagonism at disease sites, supporting depot or sustained-release research hypotheses.
Development & Formulation Notes
Stock Solutions & Handling
Prepare concentrated stocks (e.g., 1–10 mM) in sterile water or DMSO. For aqueous buffers, pH 6.5–7.5 typically preserves peptide integrity; confirm by HPLC if method development requires extended bench time. Use 0.22 µm filtration when sterility is essential. To minimize adsorption to plastics at low µM or nM levels, consider low-binding tubes and carrier protein (e.g., 0.1% BSA) per institutional SOPs.Assay Design
In migration and tube-formation assays, establish ligand-challenge curves (VEGFA, VEGFB, or PlGF) and test GNQWFI dose–response to define IC50 and maximal inhibition under your conditions. Record cell line, matrices, and growth factor lot numbers, as these variables influence outcomes.Combinations & Pathway Mapping
GNQWFI is well-suited for orthogonal combination designs with VEGFR2 inhibitors, anti-Ang/Tie2 agents, or matrix-modulating compounds to map network compensation. Include time-course sampling for phospho-readouts (e.g., p-FAK, p-AKT) to capture early pathway dynamics.Delivery Approaches in Animal Studies (research only)
Depending on model needs, evaluate intravitreal, peri-tumoral, intraperitoneal, or local depot administration, and consider biodegradable carriers (e.g., hyaluronic-based hydrogels, PLGA microspheres) to extend residence time. Calibrate exposure with PK sampling and correlate with functional angiogenesis endpoints.Quality & Documentation
Each lot is accompanied by CoA, HPLC chromatogram, MS identity, and residual solvent data (where applicable). Upon request, we can support with method notes for reconstitution, filtration, and short-term stability to accelerate lab onboarding.
Disclaimer
GNQWFI is supplied strictly for laboratory research use only. It is not a pharmaceutical, medical device, diagnostic, or veterinary product, and it is not intended for human ingestion, injection, or direct clinical use. Handle in accordance with institutional biosafety policies, wear appropriate PPE, and dispose of materials per local regulations. All performance characteristics described herein refer to preclinical research settings. End-users are solely responsible for determining suitability for their specific assays and for complying with all applicable laws and guidelines.
Keywords
GNQWFI; anti-Flt1 peptide; VEGFR1 antagonist; Flt-1 inhibitor peptide; VEGFA VEGFB PlGF blocker; angiogenesis inhibitor peptide; endothelial migration assay; tube formation assay; ocular neovascular research; oncology angiogenesis tool; asthma vascular remodeling; GMP peptide; CAS 853995-60-5; research-use-only peptide.
Packaging & Availability
Standard Packs: 1 mg, 5 mg, 10 mg; custom pack sizes upon request.
Bulk/Wholesale: Multi-gram lots with batch reservation and scheduled deliveries for long programs.
Documentation: CoA, HPLC/MS data; additional QC documentation available for audits.
Quality Assurance
Manufactured under GMP-compliant controls with validated purification and analytical methods. Routine testing includes identity (MS), purity (HPLC), residual solvents, water content, and appearance. Optional endotoxin and bioburden testing are available per project needs.
Custom Services
Custom aliquoting & labeling to align with your LIMS/ELN.
Formulation support for aqueous buffers, excipients, or carrier systems.
Stability study design for extended storage or shipment conditions.
Combination assay planning with related angiogenesis modulators.
Additional information
| Weight | 0.8 kg |
|---|---|
| Dimensions | 63 × 23 × 63 cm |
Q1. What is GNQWFI and how does it work?
GNQWFI is an anti-Flt1 hexapeptide that selectively antagonizes VEGFR1. By blocking VEGFR1 binding to VEGFA, VEGFB, and PlGF, GNQWFI reduces downstream signaling that drives endothelial migration and tube formation, enabling receptor-specific angiogenesis studies.
Q2. What research areas is GNQWFI best suited for?
GNQWFI is suited for oncology, ocular neovascular disease, asthma/airway remodeling, and fundamental angiogenesis research. It allows experiments that isolate VEGFR1-specific functions without directly inhibiting VEGFR2-dominant pathways.
Q3. How should GNQWFI be prepared and stored?
Reconstitute GNQWFI in sterile water, buffer, or DMSO to make concentrated stocks; filter if sterility is required. Store lyophilized material at –20 °C, protected from moisture and light, and avoid repeated freeze–thaw cycles for best stability.
Q4. Can GNQWFI be combined with other pathway inhibitors?
Yes. GNQWFI can be combined with VEGFR2 inhibitors, Tie2/Ang modulators, or matrix-targeting agents to explore pathway redundancy and multi-node control of angiogenesis in preclinical models.
Q5. Is GNQWFI suitable for ocular models?
GNQWFI is frequently evaluated in retinal/choroidal neovascular research where VEGFR1 signaling contributes to leakage and pathologic vessel growth. Delivery approach, dose, and formulation should be optimized per model.
Q6. What purity and documentation accompany GNQWFI?
Each lot of GNQWFI is 99.45% purity (HPLC), GMP-compliant, and shipped with a CoA, HPLC trace, and MS identity data. Extended QC documents are available upon request to meet internal audit needs.
Q7. Is GNQWFI approved for clinical use or diagnostics?
No. GNQWFI is for laboratory research use only and is not approved for human or veterinary applications, diagnostics, or therapeutic use.
Q8. Do you support wholesale orders and standing supply?
Yes. GNQWFI is available in wholesale quantities with batch reservation, scheduled releases, and lot-to-lot continuity plans for long-term studies.








Reviews
There are no reviews yet.