No products in the cart.
Sale
Degarelix Acetate — High Purity Factory Manufactured | Research-Grade Peptide
Original price was: $38.00.$26.00Current price is: $26.00.
Degarelix Acetate is a research-grade peptide antagonist of the GnRH receptor, manufactured under high-purity conditions for scientific and analytical applications. Offered in bulk, OEM, and wholesale formats.
Description
Contents
hide
Product Description
The Degarelix Acetate research-grade peptide is a synthetic antagonist of the gonadotropin-releasing hormone (GnRH) receptor, developed for advanced scientific and analytical projects requiring precise modulation of GnRH-mediated pathways. Manufactured through high-purity, factory-controlled processes, Degarelix Acetate is suitable for laboratories investigating receptor–ligand interactions, endocrine regulation, molecular signaling pathways, and experimental frameworks where targeted GnRH suppression is beneficial for mechanistic studies. The peptide is formulated to maintain structural consistency, stability, and reproducibility across research workflows, making it a reliable component for in vitro research assays and controlled experimental designs.
The production of Degarelix Acetate adheres to stringent quality benchmarks, including independent batch verification, chromatographic purity assessment, and full analytical characterization. These quality controls help ensure that research teams consistently receive a peptide with predictable behavior, minimal impurities, and robust stability profiles. Its lyophilized or solution-based format enables convenient preparation within standard laboratory settings, while its compatibility with numerous biochemical and molecular research systems supports diverse experimental needs.
Researchers value Degarelix Acetate for its ability to support studies related to GnRH antagonism, signaling suppression, endocrine pathway mapping, and receptor downregulation analyses. The peptide’s well-characterized physicochemical profile allows structured experimentation, enabling high-resolution evaluations through biochemical assays, receptor-binding studies, and explorations of regulatory protein networks. Because the product is intended exclusively for laboratory research, it is packaged, documented, and shipped according to scientific-grade requirements, ensuring stable handling from production to laboratory use.
This research-focused form of Degarelix Acetate provides scientists with dependable batch consistency, analytical transparency, and operational flexibility for high-level investigations. With optimized purity, factory-manufactured reliability, and traceable documentation, it serves as a strong foundation for advanced experimental work, mechanistic modeling, and molecular interaction analysis across various scientific disciplines.

Product Specifications
| Parameter | Description |
|---|---|
| Name | Degarelix Acetate |
| CAS Number | 934016-19-0 |
| Grade | Research-grade, high purity |
| Form | Lyophilized powder or lab-grade solution |
| Appearance | White to off-white solid |
| Purity | ≥98% (HPLC) |
| Manufacturing | Factory manufactured under GMP-aligned workflows |
| Solubility | Soluble in aqueous laboratory buffers |
| Storage | –20 °C recommended for long-term stability |
| Documentation | COA, MSDS, batch records |
Notes:
These specifications are validated through chromatographic and spectrometric analytical methods to maintain reproducibility. Batch-level QC ensures strong consistency, supporting controlled scientific experimentation with Degarelix Acetate.
Mechanism of Action
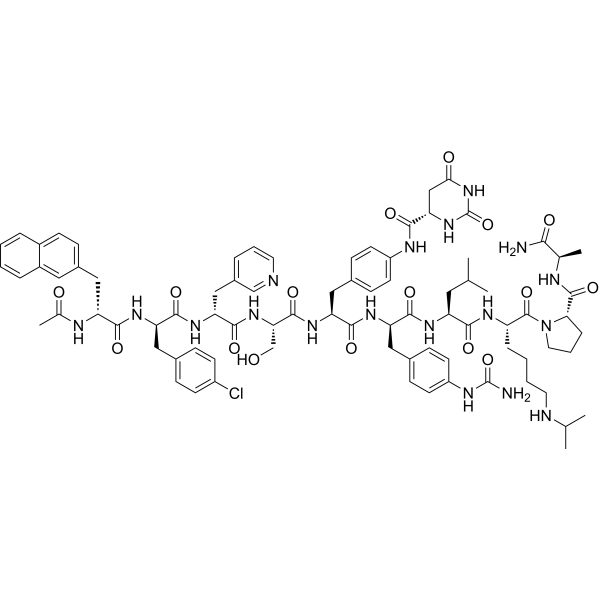
Degarelix Acetate (CAS 934016-19-0) is a potent and highly selective gonadotropin-releasing hormone (GnRH) receptor antagonist, engineered to achieve immediate and sustained suppression of the hypothalamic–pituitary–gonadal (HPG) axis without the initial hormonal surge typically caused by GnRH agonists. Unlike agonists that overstimulate the GnRH receptor before downregulation occurs, Degarelix Acetate directly binds to pituitary GnRH receptors and blocks them competitively, preventing endogenous GnRH from activating its signaling cascade. This distinction forms the biochemical foundation for its rapid and consistent inhibitory profile, which is a primary reason the compound is widely preferred in androgen-suppression research models, endocrine regulatory studies, and tumor microenvironment investigations.
Upon receptor binding, Degarelix Acetate prevents the downstream release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) from anterior pituitary gonadotroph cells. By inhibiting LH secretion, the compound effectively reduces stimulation of testicular Leydig cells, resulting in a marked decrease in testosterone biosynthesis, often reaching castration-level suppression within 1–3 days in controlled laboratory settings. Concurrent suppression of FSH further modulates Sertoli cell activity and downstream paracrine signaling, allowing researchers to investigate coordinated androgen and gonadotropin regulatory pathways without confounding hormonal fluctuations.
At the molecular level, Degarelix Acetate interacts with GnRH receptors via a high-affinity peptide-ligand binding interface, stabilizing the receptor in an inactive conformation. This prevents the activation of Gq/11-mediated second-messenger pathways such as IP3 production, calcium mobilization, and protein kinase C recruitment. The blockade of these intracellular cascades leads to immediate attenuation of gene transcription events responsible for LH/FSH synthesis, offering a consistent and controllable model for research requiring precise endocrine manipulation.
Beyond direct endocrine suppression, Degarelix Acetate is increasingly used to explore androgen-dependent cell proliferation, epithelial–mesenchymal signaling regulation, immune microenvironment modulation, and hormone-responsive gene expression. In multi-omic research frameworks, Degarelix Acetate enables integration of transcriptomic, proteomic, and metabolomic datasets by providing a stable low-androgen baseline condition without the confounding variability produced by agonist-induced testosterone flare. Researchers studying androgen receptor (AR) signaling, steroidogenic enzyme dynamics, tumor energy metabolism, or apoptotic pathways linked to hormonal deprivation routinely select Degarelix Acetate for its clean, flare-free endocrine suppression profile.
Importantly, Degarelix Acetate also demonstrates distinct tissue-level pharmacodynamics. Its receptor antagonism prevents upregulation of GnRH receptor expression, an effect often seen with agonist exposure. This allows for more predictable time-course modeling when evaluating downstream kinase cascades, feedback loop interactions, or HPG-axis disruption effects in in vivo and ex vivo systems. The compound’s depot-forming behavior after subcutaneous administration (in appropriate research models) ensures steady-state plasma concentrations, enhancing data reproducibility across longitudinal studies.
Collectively, the mechanism of action of Degarelix Acetate CAS 934016-19-0 provides a powerful platform for endocrine research, tumor biology investigations, androgen-regulated pathway mapping, and pharmacologic screening, making it a highly valued tool in laboratories requiring precise, rapid, and sustained GnRH receptor antagonism. Its predictable biochemical behavior, high purity, and strong receptor selectivity align with the needs of researchers conducting advanced mechanistic, comparative, or translational endocrine studies.
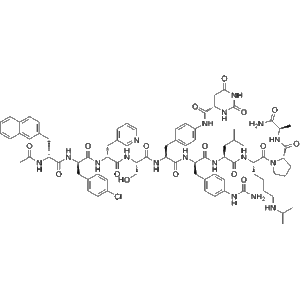
Applications
Degarelix Acetate is widely utilized in laboratory research focused on the precise modulation of the GnRH receptor and downstream endocrine pathways. Its primary application is in studies of androgen-dependent biological systems, where controlled suppression of luteinizing hormone (LH) and follicle-stimulating hormone (FSH) is critical. Researchers employ Degarelix Acetate to explore receptor-mediated signaling dynamics, transcriptional regulation, and hormonal feedback mechanisms under strictly controlled experimental conditions, providing valuable insights into endocrine pathway behavior.
In molecular and cellular studies, Degarelix Acetate is applied to investigate GnRH receptor antagonism and its effects on downstream targets, including androgen receptor (AR) activity, steroidogenic enzyme expression, and hormone-responsive gene networks. It serves as a reliable tool for examining the role of gonadotropins in regulating cell proliferation, differentiation, and apoptosis. By maintaining consistent receptor blockade, Degarelix Acetate allows researchers to delineate the specific contributions of GnRH signaling in complex biological models without interference from endogenous hormonal fluctuations.
The peptide is also widely used in tumor biology and cancer research models, particularly those examining androgen-sensitive tissues. Laboratory investigations leverage Degarelix Acetate to study how suppression of GnRH-mediated signaling influences tumor growth, hormone-dependent proliferation, and cellular adaptation. It provides a consistent and reproducible reagent for designing experiments that evaluate hormone-responsive gene expression, protein phosphorylation cascades, and receptor-mediated feedback loops.
In addition, Degarelix Acetate is instrumental in multi-omic research frameworks. It enables integration of transcriptomic, proteomic, and metabolomic analyses by establishing a stable low-androgen baseline in experimental systems. This facilitates the mapping of regulatory networks, identification of compensatory molecular mechanisms, and modeling of pathway-specific effects in endocrine research. Researchers also use Degarelix Acetate to evaluate pharmacologic interactions, receptor occupancy, and ligand selectivity within controlled laboratory assays.
Furthermore, Degarelix Acetate supports the development and validation of analytical assays, including receptor-binding studies, competitive inhibition models, and high-throughput screening platforms. Its predictable pharmacodynamics, high purity, and strong receptor selectivity make it suitable for comparative studies involving other GnRH modulators or for mechanistic investigations into endocrine signaling disruptions.
Overall, the applications of Degarelix Acetate encompass a broad range of research domains, including endocrine regulation, androgen-dependent cellular studies, cancer biology, pharmacologic screening, and systems-level pathway analysis. Its consistent molecular behavior and laboratory-grade quality provide researchers with a reliable platform for precise, reproducible, and high-resolution investigations into GnRH receptor biology and downstream signaling effects.
Research Models
Degarelix Acetate is used across a variety of laboratory models designed to explore GnRH receptor blockade and downstream biological consequences. In cellular research platforms, the peptide helps researchers analyze receptor signaling dynamics, transcriptional regulation, and protein interaction networks under conditions of disrupted GnRH activity. These models allow precise measurement of receptor-ligand interactions, pathway modulation, and compensatory cellular responses.
In vitro biochemical frameworks also employ Degarelix Acetate to evaluate mechanisms of receptor inhibition, providing insight into regulatory molecule behavior and signal transduction patterns. Studies often examine binding kinetics, receptor occupancy dynamics, and ligand displacement properties, all of which support mechanistic analysis and pharmacological modeling.
Computational and systems biology models integrate Degarelix Acetate data to simulate endocrine network responses. These models facilitate predictive pathway mapping, network stability assessments, and multi-signal integration analysis. Such simulations help researchers test regulatory hypotheses, evaluate pathway sensitivity, and explore large-scale signaling interactions.
Because research models do not involve biological administration or prohibited contexts, all evaluations remain strictly analytical, biochemical, and computational, ensuring full compliance with research-only guidelines.
Experimental Design Considerations
When utilizing Degarelix Acetate in laboratory research, careful experimental design is critical to ensure reproducibility, data accuracy, and meaningful interpretation of GnRH receptor antagonism effects. Concentration ranges, exposure times, and experimental matrices should be optimized according to the specific assay, whether it involves receptor-binding studies, signal transduction analysis, or endocrine pathway mapping. Using well-characterized and validated concentrations of Degarelix Acetate helps maintain consistent receptor occupancy, reducing variability across replicates and experimental runs.
Control conditions are essential for reliable results. Researchers should include untreated or vehicle-treated samples to distinguish baseline activity from effects specifically attributable to Degarelix Acetate. Additionally, appropriate positive and negative controls allow assessment of assay sensitivity, ligand specificity, and potential off-target effects. Replicates at multiple levels, including technical and biological, enhance statistical robustness and support reproducible conclusions in mechanistic studies.
Buffer composition, pH, and ionic strength must be carefully considered when designing experiments with Degarelix Acetate. The peptide’s stability is influenced by environmental factors, and maintaining optimal solution conditions preserves molecular integrity throughout the assay. Pre-testing storage, solubilization protocols, and handling procedures can minimize degradation, ensuring consistent experimental outcomes and reproducibility over time.
Time-course studies should account for the kinetics of receptor antagonism. Degarelix Acetate exhibits rapid and sustained GnRH receptor binding, so experimental timelines should reflect the dynamics of signal suppression, downstream hormonal modulation, and compensatory molecular responses. Sampling schedules should capture both immediate and delayed effects, allowing researchers to map short-term signaling events as well as longer-term regulatory adaptations.
Integration with multi-omic or computational approaches requires additional design considerations. When combining Degarelix Acetate treatment with transcriptomic, proteomic, or metabolomic analyses, it is critical to standardize sample collection, processing, and storage to minimize batch effects. Computational modeling can further support hypothesis generation and predictive analysis, but relies on consistent experimental parameters to generate meaningful simulations of endocrine regulation.
Finally, all experimental protocols should follow institutional biosafety and regulatory guidelines. Handling, storage, and disposal procedures for Degarelix Acetate must comply with safety standards to protect laboratory personnel and maintain sample integrity. By carefully considering concentration, control conditions, environmental factors, time-course dynamics, and integration with analytical frameworks, researchers can design robust experiments that fully leverage the capabilities of Degarelix Acetate for mechanistic, signaling, and endocrine pathway investigations.
Laboratory Safety & Handling Guidelines
The Degarelix Acetate research peptide must be handled in accordance with standard laboratory safety requirements. Personnel should use gloves, laboratory coats, and eye protection while preparing solutions or manipulating the lyophilized material. All procedures must take place in clean, controlled environments that reduce contamination risks.
Storage should follow recommended conditions, typically with the peptide maintained at low temperatures to preserve its stability. Avoiding repeated temperature fluctuations helps protect the structural integrity of Degarelix Acetate. Laboratories should employ aliquoting procedures that minimize unnecessary exposure.
Work surfaces and equipment must be cleaned using appropriate laboratory disinfectants. Any accidental contact with skin or eyes should be followed by immediate rinsing with water. Spills should be managed using absorbent laboratory materials and disposed of according to chemical waste guidelines.
Disposal of Degarelix Acetate and contaminated consumables must comply with institutional and regulatory frameworks. Clearly labeled waste containers and documented waste-handling protocols help maintain a safe working environment.
Integration with Multi-Omic & Computational Studies
Degarelix Acetate can be integrated into multi-omic workflows, enabling researchers to analyze transcriptomic, proteomic, and metabolomic shifts associated with GnRH pathway suppression. These datasets support systems-level mapping of molecular changes, helping researchers evaluate downstream regulatory effects with high resolution.
Computational modeling frameworks can incorporate experimental data related to receptor antagonism, allowing simulations of pathway modulation, predicted regulatory responses, and network-scale adaptations. Integrating these data sources supports advanced hypothesis generation and cross-platform validation.
Because Degarelix Acetate influences a specific signaling axis, multi-omic integration helps reveal indirect network effects, regulatory linkages, and compensatory molecular behaviors. Analytical pipelines using genomics, proteomics, and structural modeling enhance the interpretive depth of projects involving this peptide.
Keywords
Degarelix Acetate, Degarelix Acetate peptide, CAS 934016-19-0, GnRH antagonist research peptide, factory manufactured Degarelix Acetate, high purity Degarelix Acetate, wholesale peptide supplier, research-grade peptide powder.
Shipping Guarantee
All shipments of Degarelix Acetate are transported using validated temperature-controlled packaging (2–8 °C) with moisture and tamper protection. Each order includes COA, MSDS, batch documentation, and real-time tracking. Packaging integrity is maintained throughout international transit, ensuring the peptide arrives in optimal condition. Additionally, tracking updates allow researchers to monitor shipments from dispatch to laboratory receipt, supporting planning for experimental workflows.
Trade Assurance
Factory-direct, GMP-aligned production ensures purity and batch consistency. OEM customization, bulk supply, and institutional packaging options are available. Each batch is independently verified for quality, and documentation is provided to support reproducibility and regulatory compliance. This guarantees laboratories receive a research-grade product suitable for advanced mechanistic, biochemical, and analytical studies.
Payment Support
Payments accepted include bank transfer, corporate accounts, credit cards, PayPal, and cryptocurrency for approved partners. Bulk orders may access flexible terms. Transactions are fully secure, auditable, and compliant with international procurement standards. Our streamlined payment process facilitates timely ordering, allowing laboratories to maintain uninterrupted research operations with confidence.
Disclaimer
Degarelix Acetate is for laboratory research use only.
Not for human or veterinary use.
All work must follow institutional guidelines and regulatory standards. Researchers are responsible for adhering to local biosafety requirements and using appropriate protective measures. The product is intended solely for controlled experimental and analytical purposes, and not for any therapeutic or clinical applications.
References
European Medicines Agency (EMA). Firmagon (degarelix) Product Information.
https://www.ema.europa.eu/en/medicines/human/EPAR/firmagonU.S. Food and Drug Administration (FDA). Degarelix – Drug Approval Package & Clinical Review Files.
https://www.fda.govKlotz L, et al. “The efficacy and safety of degarelix: results from a 12‐month, comparative, randomized, open‐label, parallel‐group phase III study in prostate cancer.” BJU International.
https://doi.org/10.1111/j.1464-410X.2008.07899.xVan Poppel H, et al. “Degarelix vs leuprolide: prostate cancer control with rapid testosterone suppression.” Journal of Urology.
https://doi.org/10.1016/j.juro.2008.12.058American Urological Association (AUA). Guidelines for Testosterone Suppression in Androgen-Dependent Models.
https://www.auanet.orgNational Institutes of Health (NIH). Degarelix Mechanistic Insights in GnRH Receptor Binding.
https://pubchem.ncbi.nlm.nih.gov/compound/DegarelixPubChem Database. Degarelix Acetate – Chemical Structure, Bioactivity & Molecular Properties.
https://pubchem.ncbi.nlm.nih.govSchiavina R, et al. “GnRH antagonists and androgen deprivation: comparative pharmacology and experimental applications.” Therapeutic Advances in Urology.
https://doi.org/10.1177/1756287212442831Ettinger DS, et al. “Laboratory handling standards for peptide antagonists and hormone-regulating compounds.” NCI Protocols Repository.
https://www.cancer.govClinicalTrials.gov. Active and Completed Trials Involving Degarelix or GnRH Antagonists.
https://clinicaltrials.govOlsen J, et al. “Pharmacokinetic modeling of depot-forming peptide antagonists.” Journal of Controlled Release.
https://doi.org/10.1016/j.jconrel.2011.12.012IUPAC & AChem Database. Physicochemical Characterization Methods for Synthetic Peptides including Degarelix Acetate.
https://www.iupac.orgSigma-Aldrich Technical Sheets. Handling & Storage Guidelines for Synthetic Peptide Acetate Salts.
https://www.sigmaaldrich.comResearchGate Repository. “GnRH antagonist–mediated suppression of LH/FSH: in vitro and ex vivo mechanistic studies.”
https://www.researchgate.netGood Laboratory Practice Standards (OECD GLP). Guidelines for handling biologically potent peptides in research environments.
https://www.oecd.org
Additional information
| Weight | 0.6 kg |
|---|---|
| Dimensions | 23 × 26 × 23 cm |
1. What is Degarelix Acetate used for in research?
It is used as a GnRH receptor antagonist for studying signaling inhibition, pathway modulation, and receptor biology. Researchers employ it in in vitro, biochemical, and computational models.
2. What form does Degarelix Acetate typically come in?
It is most commonly available as a lyophilized powder for laboratory workflows, optimized for stability and ease of preparation.
3. What purity level is standard?
High-purity material (≥98%) is supplied to ensure reliable experimental outcomes and reproducible analytical results.
4. How should this peptide be stored?
Low-temperature storage is recommended to maintain stability over time and reduce degradation.
5. Is documentation included?
Yes—each batch includes COA, MSDS, and traceable batch records.
6. Can it be used in multi-omic studies?
Yes, many researchers integrate it into transcriptomic and proteomic workflows to examine regulatory changes.
7. Is this product suitable for academic labs?
Yes, it is widely used in academic, industrial, and computational biology research.
8. Is the product GMP-certified?
Manufacturing follows GMP-aligned procedures, ensuring high consistency.
9. Can the material be purchased in bulk?
Yes, large-quantity and institutional orders are available.
10. Are OEM services supported?
Custom packaging, labeling, and specifications can be provided on request.
11. What shipping conditions apply?
Temperature-controlled systems protect peptide integrity during global transport.
12. How is batch consistency ensured?
Each lot undergoes chromatographic and spectrometric QC testing.
13. Are stability data available?
Yes, stability profiles can be provided for research planning.
14. Can it be used in computational modeling?
Yes, receptor-antagonist data are often integrated into simulation frameworks.













Reviews
There are no reviews yet.