No products in the cart.
Sale
Exemestane | CAS 107868-30-4 | Steroidal Aromatase Inhibitor for Endocrine and Oncology Research
Original price was: $3.00.$2.00Current price is: $2.00.
Exemestane is a steroidal aromatase inhibitor used in laboratory and preclinical research to investigate estrogen biosynthesis suppression and endocrine regulation. It is widely applied in studies exploring hormone-dependent cell signaling, cancer metabolism, and endocrine pharmacology. As a potent irreversible aromatase inactivator, it provides valuable insights into enzymatic inhibition kinetics and steroidogenesis modulation at the molecular level.
Description
Contents
hide
Product Description
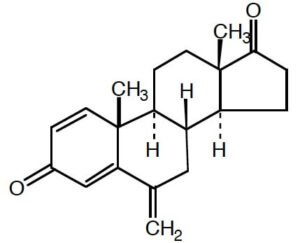
Exemestane is a synthetic steroidal compound that functions as a selective, irreversible inhibitor of the aromatase enzyme (CYP19A1). Aromatase catalyzes the final step in the biosynthesis of estrogens from androgens, and its inhibition by exemestane leads to a marked reduction in estrogen levels within biological systems. This mechanism makes it an essential molecule in preclinical investigations focused on endocrine modulation, hormone regulation, and estrogen-dependent cancer research.
Structurally, exemestane is a 6-methylene analog of androstenedione, belonging to the steroidal aromatase inhibitor class. Its molecular scaffold enables covalent binding to the heme group of aromatase, leading to irreversible enzyme inactivation. This “suicide inhibition” mechanism differentiates it from nonsteroidal inhibitors, which typically bind reversibly.
In biochemical research, exemestane is utilized to examine:
Enzyme–substrate kinetics of CYP19A1 inhibition.
Hormonal regulation of estrogen synthesis.
Metabolic pathways involving androgens and estrogens.
Cross-regulation between endocrine and metabolic signaling networks.
At the molecular level, exemestane’s interaction with aromatase results in a time-dependent reduction of catalytic turnover, providing a model for irreversible enzyme inhibition studies. It also serves as a chemical probe for elucidating the structural dynamics of P450 enzymes and their active-site conformations.
In vitro research frequently explores the effects of exemestane on gene expression associated with estrogen synthesis, receptor activation, and downstream cell signaling. Its ability to alter the transcriptional activity of estrogen-responsive genes makes it a valuable compound for studies involving hormone-responsive cell lines.
Beyond enzymology, exemestane is also investigated in metabolic and pharmacokinetic studies, including its oxidation and conjugation pathways. Analytical methods such as HPLC, LC–MS/MS, and NMR spectroscopy are used to characterize its degradation and metabolic intermediates under various experimental conditions.
Researchers value exemestane for its high specificity, stability, and reproducibility in biological assays. Its well-defined mechanism of action, combined with its irreversible binding nature, has established it as a model compound in enzymology, steroid biosynthesis research, and pharmacodynamic analysis of aromatase inhibition.
Product Specifications
| Item | Details |
|---|---|
| Product Name | Exemestane |
| CAS Number | 107868-30-4 |
| Synonyms | Aromasin; 6-Methyleneandrostenedione |
| Molecular Formula | C20H24O2 |
| Molecular Weight | 296.41 g/mol |
| Purity | ≥99% |
| Appearance | White to off-white crystalline powder |
| Solubility | Slightly soluble in water; soluble in DMSO, ethanol, and methanol |
| Storage Temperature | 2–8 °C |
| Category | Steroidal aromatase inhibitor; research-grade compound |
| Applications | Endocrine regulation, aromatase inhibition studies, estrogen biosynthesis research |
| Formulation | Suitable for in vitro and ex vivo experimental models |
| Stability | Stable under recommended storage conditions |
| Shelf Life | 24 months |
| Supplier Type | Research chemical supplier |
| Intended Use | For laboratory research use only |
Mechanism of Action
Exemestane acts as an irreversible inhibitor of the aromatase enzyme (CYP19A1), the key enzyme responsible for converting androgens (such as testosterone and androstenedione) into estrogens (estradiol and estrone). This inhibition leads to the suppression of estrogen synthesis, making it a valuable model compound for endocrine and oncology research.
1. Covalent Enzyme Inactivation
Unlike reversible inhibitors, exemestane forms a covalent bond with the aromatase active site. Its steroidal structure allows it to mimic the natural substrate androstenedione, binding to the heme moiety of the enzyme. Upon oxidation, a reactive intermediate is generated that covalently modifies the active site, leading to irreversible loss of enzymatic activity.
2. Steroidal Binding Affinity
Because exemestane shares structural similarity with endogenous steroids, it exhibits strong affinity for the substrate-binding pocket of aromatase. This feature enhances its selectivity and minimizes interactions with other cytochrome P450 enzymes, making it a precise tool for studying the molecular basis of steroid metabolism.
3. Reduction of Estrogen Biosynthesis
By inactivating aromatase, exemestane prevents the conversion of androgens into estrogens, resulting in reduced estrogenic signaling. This mechanism is essential in preclinical studies investigating hormone-dependent cell growth, estrogen receptor modulation, and downstream signaling pathways.
4. Regulation of Gene Expression
Experimental studies show that exemestane alters the transcription of estrogen-responsive genes, including those involved in cell proliferation and apoptosis. It also affects feedback loops within the hypothalamic–pituitary–gonadal axis, providing a comprehensive model for studying hormonal homeostasis.
5. Applications in Oncology and Endocrine Research
Exemestane’s unique irreversible mechanism makes it a leading compound in experimental oncology and hormone biology. It is widely used in in vitro assays, cell-based studies, and biochemical evaluations to understand aromatase function, inhibitor kinetics, and hormonal control systems.
6. Downstream Molecular Pathways
Exemestane’s suppression of estrogen biosynthesis has broad effects on signal-transduction networks. In cell-culture systems, reduced estrogen availability diminishes activation of ERα and ERβ, resulting in lower transcription of estrogen-responsive genes such as c-myc and cyclin D1. This leads to a cascade of changes in cell-cycle checkpoints, protein kinase activity, and apoptotic signaling. Such molecular outcomes are used experimentally to map the links between hormonal control and cellular metabolism.
7. Pharmacokinetic and Metabolic Research
In preclinical metabolism studies, exemestane undergoes oxidation and conjugation primarily through hepatic CYP450 pathways. Its major metabolite, 17-hydroexemestane, retains partial biological activity and is often quantified to evaluate enzyme turnover. Researchers use this metabolic profile to develop computational models of enzyme-substrate dynamics and predict irreversible inhibitor behavior.
8. Comparative Enzyme Inhibition Studies
In comparative laboratory studies, exemestane is frequently evaluated against non-steroidal aromatase inhibitors such as letrozole and anastrozole. Its distinct mechanistic profile as a “suicide inhibitor” allows researchers to contrast time-dependent versus competitive enzyme inhibition kinetics, enabling detailed structure–activity relationship analyses.
9. Cellular Model Applications
In vitro assays employ human hepatocyte, adipocyte, and breast-derived cell lines to examine exemestane’s effects on estrogen receptor expression and steroidogenic enzyme activity. Researchers measure changes in mRNA expression, protein levels, and metabolite ratios to quantify the extent of aromatase inactivation. The compound’s reliability makes it a standard reference molecule for evaluating novel aromatase inhibitors.
Side Effects
In controlled laboratory environments, Exemestane is well-tolerated at research-level concentrations. However, scientists observing cellular responses may note certain dose-dependent effects:
Cellular viability changes: High micromolar doses can induce mild cytotoxicity in estrogen-dependent cell lines, likely due to disruption of hormone-regulated metabolic processes.
Oxidative stress: Metabolic oxidation of the steroidal core may generate reactive species in vitro; researchers often co-monitor ROS markers.
Membrane interaction: Because of its lipophilic nature, exemestane can alter membrane fluidity in cell culture at elevated concentrations.
These observations are experimental and do not represent clinical outcomes. The compound is strictly intended for laboratory use and should be handled under appropriate biosafety conditions.
Keywords
Exemestane, Aromasin, CAS 107868-30-4, steroidal aromatase inhibitor, CYP19A1 research, hormone biosynthesis study, endocrine regulation, oncology research, irreversible enzyme inactivation, high-purity research compound, research chemical manufacturer, OEM and bulk production China.
Shipping Guarantee
All Exemestane shipments are handled using validated cold-chain logistics to preserve compound integrity. Each package is sealed in moisture-proof containers with secondary protective wrapping and continuous temperature monitoring. Products are shipped via express international couriers with full tracking and insurance coverage.
Trade Assurance
We Exemestane ensure product authenticity, verified ≥ 99 % purity, and compliance with analytical standards (HPLC, MS, and NMR). Each batch is supplied with a Certificate of Analysis (CoA). Our trade assurance policy guarantees replacement or refund for any deviation from listed specifications.
Payment Support
We Exemestane provide flexible and secure global payment options to support international research transactions. Accepted payment methods include PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). All transactions are protected by industry-standard encryption and verified payment gateways to ensure confidentiality and fund security.
Disclaimer
All Exemestane products listed are intended for laboratory research use only and not for human or veterinary use. They are not drugs, medical devices, or diagnostics and should not be administered to humans or animals. Researchers must handle all materials in accordance with institutional biosafety and chemical safety guidelines. The information provided is for scientific reference only and does not imply therapeutic efficacy, safety, or regulatory approval.
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
What is Exemestane used for in research?
It is used to study aromatase inhibition, estrogen biosynthesis mechanisms, and endocrine signaling pathways.
Is Exemestane a high-purity compound?
Yes, each batch is verified ≥ 99 % pure via HPLC and MS analysis.
What type of compound is it?
A steroidal aromatase inhibitor belonging to the androstenedione analog class.
How should Exemestane be stored?
Store between 2–8 °C, protected from light and moisture.
What is its appearance?
White to off-white crystalline powder.
Is it soluble in water?
Slightly soluble in water; readily soluble in organic solvents like DMSO and ethanol.
Do you provide a Certificate of Analysis (CoA)?
Yes, each shipment includes a CoA verifying purity and identity.
Can it be used for cell-based assays?
Yes, it is commonly used in in vitro hormone regulation and enzyme inhibition studies.
Is bulk or OEM production available?
Yes, OEM and bulk production services for research institutions are available.
What is the minimum order quantity?
MOQ varies by batch; both small and bulk quantities are supported.
Where is the product manufactured?
Produced in our certified China facility specializing in research-grade chemicals.
How is cold-chain shipping ensured?
Temperature-controlled packaging with continuous monitoring and tracking.
Does the compound contain additives or preservatives?
No, it is supplied in pure form without stabilizers.
Can international researchers order directly?
Yes, we support direct international orders with full documentation and tracking.
Why choose your supplier for Exemestane?
We are a factory peptide supplier and research chemical manufacturer offering high-purity compounds, competitive pricing, and reliable OEM & bulk production from China.















Reviews
There are no reviews yet.