No products in the cart.
Ezatiostat — Purity 99.71% raw material
$2.00
Ezatiostat (TLK199) is a small molecule glutathione S-transferase P1-1 (GSTP1-1) inhibitor, widely used in in vitro mechanistic studies of GSTP1-mediated signaling pathways, oxidative stress modulation, and apoptosis regulation. This high-purity compound is designed for cell-free assays, biochemical analyses, and molecular mechanism research.
Factory-manufactured in China, low-cost wholesale supply supports global laboratory research.
Description
Product Description
Ezatiostat (TLK199) is a synthetic small molecule that functions as a specific inhibitor of glutathione S-transferase P1-1 (GSTP1-1), a key regulator of cellular redox balance and apoptosis signaling. It has been extensively used in in vitro research to study GSTP1-mediated pathways, oxidative stress responses, and protein S-glutathionylation mechanisms. Its chemical stability and defined structure make it suitable for biochemical assays, enzyme kinetics, and molecular mechanism studies without in vivo or clinical use.
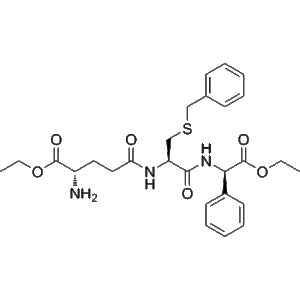
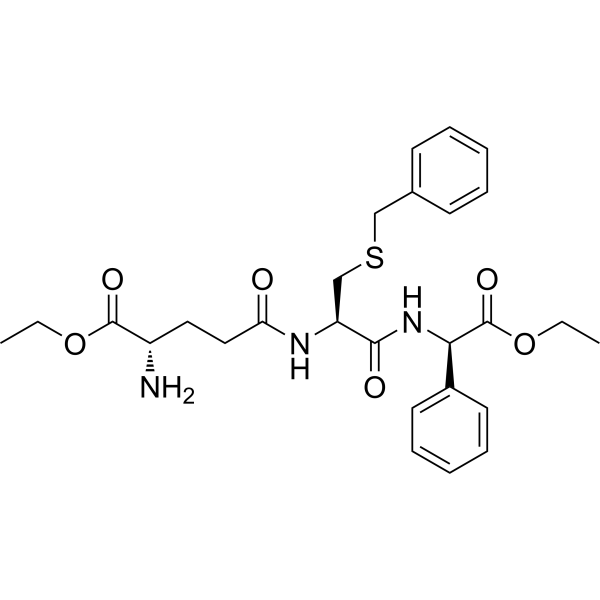
Chemically, Ezatiostat contains a tripeptide mimic motif that selectively binds the active site of GSTP1, disrupting its interaction with target proteins such as c-Jun N-terminal kinase (JNK). By inhibiting GSTP1, Ezatiostat facilitates the study of stress-induced apoptotic signaling, enzyme-substrate interactions, and redox-mediated regulatory mechanisms.
Key Features and Advantages:
High Purity: ≥99% verified by HPLC ensures reproducible mechanistic studies and minimal interference from impurities.
In Vitro Research Applications: Ideal for GSTP1 inhibition studies, apoptosis assays, oxidative stress pathway analysis, and biochemical characterization.
Chemical Stability: Stable under standard laboratory conditions, suitable for extended experimental workflows.
Mechanistic Insight: Enables investigation of protein S-glutathionylation, kinase activation, and redox signaling networks.
Factory-Direct Supply: Manufactured in China, available for bulk and wholesale purchase, ensuring consistent availability for long-term research.
Experimental Applications:
GSTP1 Enzyme Inhibition: Quantify enzymatic activity reduction and evaluate inhibitor kinetics in cell-free or purified enzyme systems.
Apoptosis and Redox Signaling: Study the effects of GSTP1 inhibition on JNK activation, downstream apoptotic pathways, and oxidative stress regulation.
Molecular Mechanism Analysis: Assess GSTP1 interactions with substrates, inhibitors, and regulatory proteins in vitro.
Computational and Structural Studies: Ezatiostat’s defined chemical structure allows docking and molecular dynamics simulations to model GSTP1-ligand interactions and binding affinities.
Ezatiostat is intended strictly for in vitro molecular and mechanistic studies, providing a highly reproducible platform for exploring GSTP1 biology, oxidative stress pathways, and protein regulatory networks.
Product Specifications
| Parameter | Specification |
|---|---|
| Product Name | Ezatiostat (TLK199) |
| Synonyms | TLK199, Ezatiostat hydrochloride |
| CAS Number | 220899-65-0 |
| Molecular Formula | C₂₂H₃₄N₄O₆S |
| Molecular Weight | 474.6 g/mol |
| Purity | ≥99% (HPLC or equivalent) |
| Appearance | White to off-white powder |
| Solubility | DMSO ≥50 mg/mL; water limited solubility, ultrasonication recommended |
| Storage | Dry, protected from light, -20°C |
| Quality Control | COA and MSDS provided; batch-to-batch consistency guaranteed |
| Stability | Stable under standard laboratory conditions for prolonged storage |
Detailed Notes:
High Purity & Validation: Ensures reproducible results in GSTP1 inhibition and apoptosis assays.
Solubility: Primarily soluble in DMSO for in vitro assays; use ultrasonication for higher concentrations.
Structural Integrity: Maintains chemical and conformational stability critical for enzyme binding studies.
Batch Traceability: Factory-manufactured in China with COA/MSDS, supporting bulk research applications.
Laboratory Compatibility: Suitable for enzyme kinetics, oxidative stress modeling, and mechanistic molecular studies.
Mechanism of Action
Ezatiostat is a selective inhibitor of GSTP1-1, an enzyme that regulates cellular redox balance and apoptosis signaling. Its tripeptide-mimic motif binds the active site of GSTP1, preventing interaction with target proteins such as JNK, thus modulating downstream apoptotic and stress-response pathways.
GSTP1 Inhibition
Ezatiostat binds to GSTP1, blocking its enzymatic activity and inhibiting conjugation of glutathione to electrophilic substrates.
Disruption of GSTP1 activity leads to activation of stress kinases such as JNK in in vitro assays.
Redox and Apoptotic Pathways
By inhibiting GSTP1, Ezatiostat allows accumulation of reactive oxygen species (ROS) in controlled studies, enabling investigation of oxidative stress mechanisms.
Facilitates mechanistic studies of protein S-glutathionylation and stress-activated apoptotic signaling.
Molecular Interaction Dynamics
Ezatiostat demonstrates high binding specificity and affinity for GSTP1 in cell-free and purified enzyme systems.
Supports kinetic studies, including enzyme inhibition constants and substrate competition analysis.
In Vitro Applications
GSTP1 enzymatic activity assays
JNK phosphorylation and apoptosis modeling
Oxidative stress and ROS generation studies
Computational docking and molecular dynamics simulations to predict binding and inhibitor efficiency
Ezatiostat’s mechanism of action is defined by GSTP1 inhibition, modulation of redox-sensitive signaling pathways, and promotion of apoptotic signaling in controlled experimental systems.
Applications
Ezatiostat (TLK199) is a versatile research-grade compound designed for in vitro molecular mechanism studies, particularly in the context of GSTP1 inhibition, apoptosis regulation, and oxidative stress signaling. Its high purity and structural stability make it suitable for diverse laboratory applications that explore enzyme function, signaling pathways, and redox modulation in controlled experimental conditions.
In Vitro Enzyme Assays
Ezatiostat is widely used in GSTP1 enzymatic inhibition studies to evaluate enzyme kinetics, substrate specificity, and inhibitor potency. Researchers can measure enzyme activity reduction and assess mechanistic effects on downstream signaling pathways, such as JNK activation and apoptosis induction.
Oxidative Stress and Apoptosis Research
By inhibiting GSTP1, Ezatiostat facilitates the study of reactive oxygen species (ROS) accumulation, S-glutathionylation, and redox-sensitive cellular responses. This allows detailed mechanistic exploration of apoptotic signaling, oxidative stress regulation, and stress kinase activation in cell-free or purified protein models.
Molecular Mechanism and Structural Studies
Ezatiostat supports enzyme-ligand interaction studies, protein-protein binding investigations, and computational docking simulations. Its defined chemical structure enables molecular modeling, prediction of binding conformations, and analysis of structure-activity relationships (SAR).
Radiolabel-Free Mechanistic Applications
While Ezatiostat is non-radioactive, it is compatible with fluorescent or labeled substrates for kinetic and mechanistic assays. Researchers can investigate GSTP1-mediated pathways using high-precision in vitro detection methods without involving in vivo systems.
Multi-Disciplinary Research Integration
Ezatiostat’s reproducibility and high specificity make it suitable for integration with proteomics, metabolomics, and bioinformatics pipelines, enabling a comprehensive understanding of GSTP1 function, redox regulation, and apoptotic mechanisms.
In summary, Ezatiostat provides a reliable platform for in vitro studies of GSTP1 inhibition, redox signaling, apoptosis, and enzyme mechanistic analysis, making it an essential tool for biochemical, molecular, and computational research.
Research Models
Ezatiostat (TLK199) is designed for controlled in vitro research, enabling reproducible studies of GSTP1 inhibition, oxidative stress regulation, and apoptosis signaling pathways. Its chemical stability, high purity, and well-defined structure make it compatible with a variety of experimental models for mechanistic and molecular investigations.
Purified Enzyme Systems
Ezatiostat can be applied to purified GSTP1 protein assays, allowing researchers to quantify enzyme inhibition kinetics, substrate competition, and binding affinity. These systems enable precise evaluation of inhibitor potency and mechanistic insight into GSTP1-mediated redox regulation.
Cell-Free and Membrane-Based Models
Cell-free systems, including cytosolic extracts and recombinant protein preparations, are used to study ROS accumulation, S-glutathionylation, and apoptosis signaling under defined conditions. Ezatiostat allows detailed mechanistic assessment without interference from other cellular components.
Computational and Structural Models
The defined chemical structure of Ezatiostat supports molecular docking, binding simulation, and molecular dynamics studies. Researchers can model GSTP1-inhibitor interactions, predict binding sites, and investigate structure-activity relationships (SAR) to guide mechanistic interpretations.
Multi-Omics Compatible Models
Ezatiostat is suitable for integration with proteomics and metabolomics pipelines to correlate GSTP1 inhibition with protein modifications, stress response pathways, and redox signaling profiles. This multi-layered approach enhances understanding of mechanistic pathways and cellular outcomes.
Integrated Experimental Approaches
By combining purified enzyme assays, cell-free models, and computational simulations, researchers can obtain a comprehensive mechanistic profile of GSTP1 inhibition. Ezatiostat’s reproducibility, stability, and high affinity ensure reliable results across multiple experimental platforms.
In summary, Ezatiostat supports enzyme-based, cell-free, computational, and multi-omic research models, making it an indispensable tool for laboratories conducting advanced in vitro mechanistic studies on GSTP1, oxidative stress, and apoptosis pathways.
Experimental Design Considerations
When using Ezatiostat (TLK199) in in vitro mechanistic studies, careful planning is essential to ensure reproducibility, accurate GSTP1 inhibition assessment, and reliable oxidative stress or apoptosis measurements. Its high purity, chemical stability, and selective activity provide a robust foundation for controlled experiments.
Enzyme Kinetics and Inhibition Studies
Concentration Optimization: Select inhibitor concentrations based on GSTP1 expression or activity levels to avoid oversaturation or underestimation of inhibitory effects.
Incubation Conditions: Maintain consistent temperature, pH, and buffer composition to preserve compound stability and enzymatic activity.
Controls and Replicates: Include negative controls, vehicle controls, and replicate measurements to validate specificity and reproducibility.
Oxidative Stress and Apoptosis Assays
ROS Measurement: Plan the timing and method for ROS detection to accurately capture oxidative stress dynamics.
Downstream Signaling Assessment: Monitor markers such as JNK activation, protein S-glutathionylation, and caspase cleavage to evaluate mechanistic outcomes.
Substrate Selection: Use well-characterized substrates or reporters compatible with Ezatiostat’s mechanism of GSTP1 inhibition.
Multi-Model Integration
Combine purified enzyme assays, cell-free systems, and computational simulations for cross-validation of mechanistic findings.
Integrate results with proteomic or metabolomic readouts to enhance mechanistic understanding of GSTP1 function and redox signaling.
Handling and Stability Considerations
Ensure proper solubilization in DMSO or other appropriate solvents and minimize freeze-thaw cycles to preserve compound integrity.
Adhere to standard laboratory practices to avoid contamination and degradation.
These design considerations facilitate robust, reproducible, and interpretable in vitro studies, providing reliable mechanistic insights into GSTP1 inhibition, oxidative stress, and apoptosis pathways.
Laboratory Safety & Handling Guidelines
Ezatiostat (TLK199) is a high-purity synthetic compound intended solely for in vitro research. Proper handling is essential to maintain compound integrity and ensure researcher safety.
General Handling
Always use personal protective equipment (PPE), including gloves, lab coats, and safety glasses.
Handle the compound in a controlled laboratory environment according to standard chemical safety protocols.
Avoid direct skin or eye contact, ingestion, and inhalation. In case of accidental exposure, follow institutional safety procedures and seek medical attention if necessary.
Storage
Store Ezatiostat in dry, light-protected containers at -20°C to maintain chemical stability and activity.
Avoid repeated freeze-thaw cycles to prevent degradation.
Clearly label all containers and keep away from incompatible substances.
In Vitro Experimental Use
Use only in enzyme inhibition, oxidative stress, apoptosis, or mechanistic biochemical assays.
Minimize contamination and ensure solutions are freshly prepared for experimental reproducibility.
When integrating with multi-omics or computational studies, maintain clean handling practices to avoid experimental artifacts.
Waste Disposal
Dispose of Ezatiostat according to local chemical waste regulations.
Do not release into the environment or dispose of with general laboratory waste.
Following these guidelines ensures the integrity of experiments while protecting researchers and maintaining compliance with laboratory safety standards.
Integration with Multi-Omic & Computational Studies
Ezatiostat (TLK199) is highly compatible with integrative in vitro research, enabling the combination of multi-omic analyses and computational modeling to explore GSTP1 inhibition, redox regulation, and apoptosis pathways. Its chemical stability and high purity support reliable experimental outcomes across diverse platforms.
Multi-Omic Integration
Proteomics: Ezatiostat allows investigation of GSTP1-associated protein networks, post-translational modifications (such as S-glutathionylation), and stress-responsive signaling proteins.
Metabolomics: Enables assessment of redox state, glutathione conjugation dynamics, and oxidative stress markers in cell-free and enzyme-based systems.
Data Correlation: The reproducible inhibitory profile facilitates correlation of enzyme activity changes with proteomic or metabolomic outputs, providing deeper mechanistic insights.
Computational Studies
Molecular Docking: Ezatiostat’s defined tripeptide-mimic structure allows accurate docking simulations with GSTP1, predicting binding sites and interaction energies.
Molecular Dynamics: Enables simulation of inhibitor-enzyme interactions over time, providing insight into binding stability, flexibility, and mechanistic pathways.
Structure-Activity Relationship (SAR) Analysis: Integration of computational predictions with experimental data supports the optimization of inhibitor design and mechanistic exploration.
By combining experimental multi-omic approaches with computational modeling, Ezatiostat provides a robust platform for mechanistic research on GSTP1 function, oxidative stress, and apoptosis signaling, facilitating advanced molecular studies and rational inhibitor development.
Keywords
Ezatiostat, TLK199, GSTP1 inhibitor, glutathione S-transferase P1-1, in vitro GSTP1 studies, apoptosis pathway research, oxidative stress modulation, enzyme inhibition assay, molecular mechanism research, high-purity research compound, mechanistic signaling studies, peptide-mimic inhibitor, redox pathway analysis, cellular stress in vitro, protein S-glutathionylation research
Payment Support
Ezatiostat can be purchased via multiple secure payment methods, facilitating global laboratory procurement:
Credit Card — Visa, MasterCard, and American Express accepted.
Telegraphic Transfer (TT / SWIFT) — Bank transfer for bulk or wholesale orders.
Cryptocurrency — Secure, encrypted payment options for international transactions.
Alipay / WeChat Pay — Available for China-based buyers.
All transactions are encrypted and verified, ensuring secure procurement for research laboratories.
Shipping Guarantee
Global Delivery: Reliable shipping for laboratory-grade reagents worldwide.
Temperature-Controlled Logistics: Ensures Ezatiostat maintains structural stability during transit.
Secure Packaging: Protects compound integrity and prevents contamination.
Tracking Available: Full shipment tracking and estimated delivery updates provided.
Trade Assurance
Factory-Direct Supply: Ezatiostat manufactured in China under strict quality control.
Batch Traceability: COA and MSDS provided for each shipment.
Bulk and Wholesale Options: Supports long-term laboratory studies and high-throughput experiments.
Verified Quality: Consistent analytical validation across batches ensures reproducibility.
Disclaimer
Ezatiostat (TLK199) is a research-grade chemical intended exclusively for in vitro molecular and mechanistic studies. It is not for human or veterinary use, ingestion, injection, or clinical application. Researchers must follow all institutional safety, handling, and disposal regulations.
References — Ezatiostat
Ezatiostat and GSTP1 Mechanism — Molecular mechanism of GSTP1 inhibition and apoptotic signaling in vitro. (PMC6374379)
Ezatiostat Enzyme Kinetics Study — Biochemical characterization of TLK199 as a GSTP1 inhibitor. (PubChem)
GSTP1 and Redox Signaling — Overview of GSTP1-mediated redox regulation and S-glutathionylation. (MDPI)
JNK Activation by GSTP1 Inhibition — Mechanistic insights from in vitro studies using TLK199. (ACS Publications)
Ezatiostat Structural and Mechanistic Data — Detailed chemical structure and in vitro activity profile. (MedChemExpress)
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
What is Ezatiostat (TLK199)?
Ezatiostat is a synthetic small-molecule GSTP1 inhibitor used in in vitro mechanistic studies, focusing on oxidative stress regulation, apoptosis, and redox signaling pathways.
Can Ezatiostat be used in vivo or clinically?
No. Ezatiostat is strictly for laboratory research and in vitro molecular studies. It is not intended for human or animal use.
What is the chemical purity of Ezatiostat?
The compound is ≥99% pure, verified by HPLC or equivalent analytical techniques, ensuring reliable reproducibility for enzyme inhibition and redox assays.
What solvents are recommended for Ezatiostat?
Ezatiostat is primarily soluble in DMSO (≥50 mg/mL). Water solubility is limited, and ultrasonication is recommended for higher concentration solutions.
How should Ezatiostat be stored?
Store in dry, light-protected containers at -20°C. Avoid repeated freeze-thaw cycles to maintain chemical stability and experimental consistency.
What are the primary research applications of Ezatiostat?
GSTP1 enzyme inhibition studies
Redox and oxidative stress pathway analysis
Apoptosis signaling mechanism investigations
Protein S-glutathionylation assays
Computational docking and molecular dynamics modeling
Is Ezatiostat compatible with purified enzyme systems?
Yes. It can be applied to purified GSTP1 protein assays to quantify inhibition kinetics, substrate competition, and binding affinity.
Can Ezatiostat be used in cell-free systems?
Yes. Ezatiostat is compatible with cytosolic extracts and recombinant protein preparations to study ROS accumulation, enzyme inhibition, and downstream signaling pathways.
Is Ezatiostat suitable for multi-omics studies?
Yes. It integrates with proteomics and metabolomics pipelines, enabling correlation of GSTP1 inhibition with protein modifications, stress response networks, and redox state analyses.
Can Ezatiostat be used for computational modeling?
Yes. Its defined chemical structure allows molecular docking, binding energy calculations, and molecular dynamics simulations to predict enzyme-inhibitor interactions.
Are bulk and wholesale purchases available?
Yes. Ezatiostat is available for factory-direct bulk procurement, suitable for long-term laboratory research projects.
What payment methods are supported?
Credit cards, TT/SWIFT transfers, cryptocurrency, Alipay, and WeChat Pay are accepted for global and China-based laboratories.
Does Ezatiostat come with batch documentation?
Yes. Each batch includes Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS) to ensure reproducibility and traceability.
What experimental controls are recommended?
Include vehicle controls, negative controls, and replicate assays to validate GSTP1 inhibition specificity and reproducibility.
Where can authoritative references for Ezatiostat be found?
Authoritative sources include PubChem, PMC articles, ACS publications, MedChemExpress data, and peer-reviewed redox/apoptosis studies.












Reviews
There are no reviews yet.