No products in the cart.
Sale
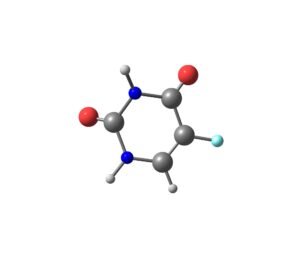
Fluorouracil | CAS 51-21-8 | Antimetabolite for Nucleic Acid Synthesis and Cytotoxicity Research
Original price was: $3.00.$2.00Current price is: $2.00.
Fluorouracil (5-FU) is a pyrimidine analog and antimetabolite widely used in preclinical research to study DNA/RNA synthesis inhibition, cell-cycle regulation, cytotoxicity pathways, apoptosis, and metabolic activation in cancer biology and biochemical assays.
Description
Product Description
Fluorouracil (5-FU), CAS 51-21-8, is a fluorinated pyrimidine analog widely applied in laboratory research for DNA replication stress, RNA misincorporation, apoptosis signaling, metabolic activation, and cytotoxic mechanism studies. Its structural similarity to uracil allows incorporation into DNA and RNA, leading to multiple cellular effects that are widely exploited in preclinical models.
Metabolic Activation and Mechanistic Metabolites
FdUMP: Inhibits thymidylate synthase (TS), causing dTMP depletion, DNA synthesis arrest, and DNA strand breaks.
FUTP: Incorporated into RNA, disrupting rRNA, tRNA, and mRNA processing, affecting translation fidelity.
FdUTP: Misincorporated into DNA, triggering mismatch repair and DNA damage signaling.
These metabolites enable 5-FU to serve as a versatile tool for studying DNA replication stress, checkpoint activation, DNA repair pathways (MMR, BER, HR), and apoptotic signaling.
Cellular Stress Responses
5-FU induces S-phase arrest, mitochondrial depolarization, ROS generation, caspase activation, and transcriptional stress. Researchers utilize these responses to examine replication stress, checkpoint activation, and DNA repair fidelity.
Combination and In Vivo Studies
Combination Studies: Paired with kinase inhibitors, nucleoside analogs, or oxidative stress modulators to evaluate synergistic or antagonistic effects.
In Vivo Studies: Applied in preclinical tumor xenografts to assess tumor growth inhibition, apoptosis induction, pharmacokinetics, and enzyme activity.
Product Specifications
| Parameter | Specification / Data |
|---|---|
| Chemical Name / Synonyms | Fluorouracil; 5-FU; Pyrimidine analog; Antimetabolite; C4H3FN2O2 |
| CAS Number | 51-21-8 |
| Molecular Formula | C4H3FN2O2 |
| Molecular Weight | 130.08 g/mol |
| Purity / Assay | ≥99% |
| Appearance | White to off-white crystalline powder |
| Solubility | Soluble in DMSO, ethanol; moderate aqueous solubility |
| Melting Point | 282–283 °C |
| Storage Temperature | 2–8 °C |
| Stability | Stable under refrigeration; protect from light and moisture |
| Analytical Methods | HPLC, MS, NMR verified; batch consistency confirmed |
| Mechanistic Target | Thymidylate synthase, RNA misincorporation, apoptosis induction, S-phase arrest |
| Structural Category | Pyrimidine analog antimetabolite |
| Applications | DNA/RNA synthesis inhibition studies, apoptosis research, cell-cycle regulation, metabolic activation research, drug resistance modeling |
| Batch Consistency | Verified by HPLC, MS, NMR |
| Regulatory Notes | For laboratory research use only |
| Origin | Factory peptide/small-molecule supplier, China peptide manufacturer, OEM & bulk production available |
| Additional Data | SMILES: C1=CN(C=O)C(=O)N=C1F; CoA provided for each batch |
Mechanism of Action
Fluorouracil exerts its preclinical research effects through thymidylate synthase inhibition, RNA misincorporation, and apoptosis induction.
Thymidylate Synthase Inhibition (FdUMP)
FdUMP forms a covalent ternary complex with TS and 5,10-methylenetetrahydrofolate, causing dTMP depletion. This triggers DNA replication arrest, strand breaks, and activation of ATR/ATM signaling, CHK1/CHK2 phosphorylation, and p53-dependent checkpoints.
RNA Misincorporation (FUTP)
FUTP incorporation into RNA disrupts rRNA, tRNA, and mRNA processing, ribosome biogenesis, and translation fidelity. Leads to ribosomal stress and unfolded protein response.
DNA Misincorporation (FdUTP)
FdUTP misincorporation activates DNA repair pathways, replication fork stalling, and apoptosis. Enables study of mismatch repair, base excision repair, and DNA damage signaling.
Apoptosis Induction
Mitochondrial depolarization, cytochrome c release, caspase-3 and -9 activation, and ROS-mediated oxidative stress. Apoptosis occurs via p53-dependent and independent mechanisms.
Drug Resistance Modeling
TS overexpression, increased DPD activity, and altered nucleotide metabolism are explored using 5-FU to understand chemoresistance mechanisms.
Side Effects
In laboratory models, Fluorouracil exhibits dose-dependent cytotoxicity:
Cellular Effects
S-phase arrest and DNA damage accumulation
ROS-mediated oxidative stress
RNA misincorporation disrupting translation and ribosomal function
Apoptotic Pathways
Caspase-3 and -9 activation
Mitochondrial depolarization and cytochrome c release
p53-dependent and independent apoptosis
Metabolic Considerations
DPD activity and nucleotide pool variability influence cytotoxicity
Used to investigate adaptive responses and combination therapy mechanisms
Laboratory Handling
Use PPE, fume hoods, and containment protocols
Strictly for research use; cytotoxic in nature
Keywords
Fluorouracil, 5-FU, pyrimidine analog, antimetabolite, thymidylate synthase inhibitor, DNA synthesis inhibitor, RNA misincorporation, apoptosis research, cytotoxicity studies, cell-cycle regulation, metabolic activation, drug resistance modeling, high-purity research chemical, factory peptide/small-molecule supplier, China peptide manufacturer, OEM & bulk production, laboratory research reagent
Shipping Guarantee
All Fluorouracil shipments of Fluorouracil are handled using validated cold-chain logistics to preserve compound integrity. Each package is sealed in moisture-proof containers with secondary protective wrapping and continuous temperature monitoring. Products are shipped via express international couriers with full tracking and insurance coverage.
Trade Assurance
We Fluorouracil ensure product authenticity, verified ≥99% purity, and compliance with analytical standards (HPLC, MS, and NMR). Each batch is supplied with a Certificate of Analysis (CoA). Our trade assurance policy guarantees replacement or refund for any deviation from listed specifications.
Payment Support
We Fluorouracil provide flexible and secure global payment options to support international research transactions. Accepted payment methods include PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). All transactions are protected by industry-standard encryption and verified payment gateways to ensure confidentiality and fund security.
Disclaimer
Fluorouracil is supplied for laboratory research purposes only. It is not intended for human or animal use, diagnostic applications, or clinical treatment. Researchers must follow all institutional and governmental safety guidelines when handling this compound. The supplier assumes no liability for misuse, unauthorized administration, or off-label applications.
References
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 18 × 16 cm |
1. What is Fluorouracil used for in laboratory research?
Fluorouracil (5-FU) is used to study DNA and RNA synthesis inhibition, apoptosis pathways, S-phase arrest, cytotoxicity mechanisms, and metabolic activation in preclinical research. It is widely applied in cancer cell biology models and mechanistic studies.
2. What is the purity of Fluorouracil supplied?
Fluorouracil is supplied at ≥99% purity, verified using HPLC, MS, and NMR to ensure consistency and reliability for laboratory experiments.
3. How should Fluorouracil be stored?
Fluorouracil should be stored as a dry powder at 2–8 °C, protected from light and moisture. Proper storage maintains stability and preserves its high purity for laboratory research.
4. Is Fluorouracil suitable for combination studies?
Yes. Fluorouracil is commonly used in combination mechanistic studies with kinase inhibitors, nucleoside analogs, or oxidative stress modulators to investigate synergistic or antagonistic effects in preclinical models.
5. Can Fluorouracil be used to study drug resistance mechanisms?
Yes. 5-FU is used to study adaptive cellular responses such as thymidylate synthase overexpression, altered dihydropyrimidine dehydrogenase (DPD) activity, and nucleotide metabolism changes in drug resistance modeling.
6. Is Fluorouracil available from a factory peptide/small-molecule supplier?
Yes. Our Fluorouracil is sourced from a reputable factory peptide/small-molecule supplier and China peptide manufacturer, with OEM and bulk production available for laboratory use.
7. What analytical methods verify Fluorouracil batches?
Each batch is verified by HPLC, MS, and NMR, ensuring batch-to-batch consistency and high-purity standards suitable for research applications.
8. Can Fluorouracil be purchased in bulk for research?
Yes. Bulk orders and OEM production are available, providing high-purity laboratory-grade Fluorouracil for preclinical studies.
9. How does Fluorouracil induce apoptosis in cells?
Fluorouracil induces apoptosis through multiple pathways including mitochondrial depolarization, ROS generation, caspase-3 and -9 activation, and both p53-dependent and independent mechanisms, which are commonly studied in mechanistic research.
10. Are high-purity Fluorouracil powders available for research use?
Yes. Our Fluorouracil powders are ≥99% high purity, specifically designed for laboratory and preclinical research purposes.
11. Can Fluorouracil be used to study RNA misincorporation?
Yes. The FUTP metabolite of 5-FU can incorporate into RNA, allowing studies on ribosomal stress, mRNA translation fidelity, and unfolded protein response pathways in vitro.
12. Does Fluorouracil require special handling for lab use?
Yes. Fluorouracil is cytotoxic and should be handled using PPE, fume hoods, and standard laboratory safety protocols. It is strictly for research use and not intended for human or animal administration.
13. Is Fluorouracil a China peptide manufacturer product?
Yes. It is sourced from a reliable China peptide/small-molecule manufacturer, ensuring consistent high-purity supply and batch quality verification.
14. Can Fluorouracil support cell-cycle and checkpoint studies?
Yes. 5-FU causes S-phase arrest and activates cell-cycle checkpoints, making it ideal for studying DNA replication stress, checkpoint mechanisms, and DNA repair pathways in preclinical models.
15. What applications are suitable for high-purity Fluorouracil?
High-purity Fluorouracil (≥99%) is suitable for DNA/RNA synthesis inhibition studies, apoptosis research, cytotoxicity evaluation, cell-cycle checkpoint analysis, metabolic activation studies, and drug resistance modeling in laboratory research.













Reviews
There are no reviews yet.