No products in the cart.
Sale
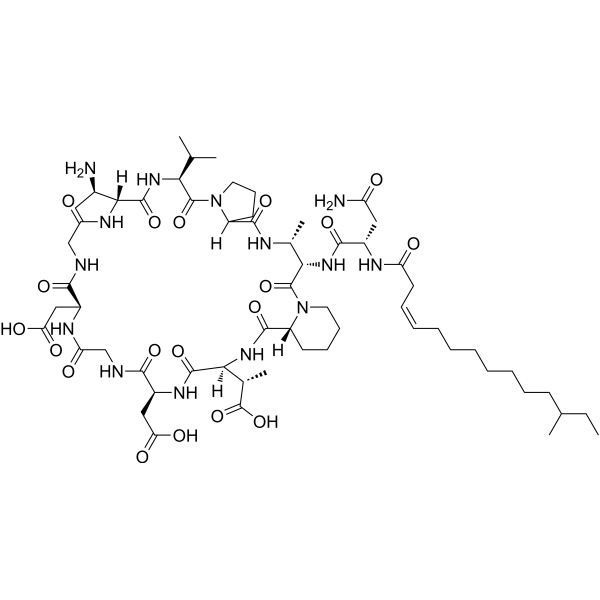
Friulimicin D (CAS 302327-42-0) | GMP Supplier & Manufacturer for Research Use
Original price was: $18.00.$15.00Current price is: $15.00.
Friulimicin D is a lipopeptide antibiotic structurally related to Friulimicin B, isolated from Actinoplanes friuliensis. With potent activity against Gram-positive bacteria, Friulimicin D is suitable for preclinical antimicrobial research, mechanism-of-action studies, and antibiotic development assays.(Please contact our staff to place an order or learn about product wholesale prices, specifications, uses and lists)
Description
Contents
hide
Product Description
Introduction
Friulimicin D is a naturally occurring lipopeptide antibiotic, produced by the actinomycete Actinoplanes friuliensis. Similar to Friulimicin B, this antibiotic exhibits a unique mechanism of antibacterial activity that distinguishes it from other lipopeptides. It is highly valuable in antimicrobial research, especially in studies focusing on Gram-positive pathogens, cell wall biosynthesis inhibition, and novel antibiotic discovery.
Chemical and Structural Properties
Friulimicin D is composed of a cyclic peptide backbone conjugated with a lipid tail, a structural feature critical for membrane interaction and bactericidal activity. The amphipathic nature of Friulimicin D allows it to integrate into bacterial membranes, disrupting essential processes while sparing eukaryotic cells under controlled experimental conditions.
Key chemical attributes:
Molecular class: Lipopeptide antibiotic
Source: Actinoplanes friuliensis
Structural similarity: Friulimicin B, with minor modifications affecting potency and spectrum
Research Relevance
The rise of antimicrobial resistance has renewed interest in lipopeptide antibiotics. Friulimicin D serves as an experimental tool for understanding bacterial membrane targeting, peptidoglycan biosynthesis inhibition, and structure–activity relationships in cyclic lipopeptides.
Applications include:
Studying bactericidal mechanisms against Staphylococcus, Enterococcus, and other Gram-positive species
Evaluating cell wall biosynthesis inhibitors in in vitro and in vivo models
Screening for synergistic interactions with other antimicrobial compounds
Preclinical testing for novel antibiotic development
Purity and Quality
Friulimicin D is supplied as a lyophilized powder with ?98% purity verified by HPLC. Manufactured under GMP-compliant processes, it ensures high reproducibility and reliability in research experiments.
Comparative Advantage
Compared to other lipopeptides, Friulimicin D offers:
Strong activity against multidrug-resistant Gram-positive strains
Minimal cytotoxicity in mammalian cell lines
Consistent lot-to-lot quality from GMP suppliers
A chemically stable structure amenable to experimental manipulation
Product Specifications
| Parameter | Details |
|---|---|
| Product Name | Friulimicin D |
| Synonyms | None commonly used; structurally related to Friulimicin B |
| CAS Number | 302327-42-0 |
| Molecular Type | Lipopeptide antibiotic |
| Molecular Formula | Available upon request |
| Molecular Weight | Available upon request |
| Appearance | White to off-white lyophilized powder |
| Purity | ? 98% (HPLC verified) |
| Solubility | Soluble in DMSO, methanol, and aqueous buffers |
| Stability | Stable for ?24 months at recommended storage conditions |
| Storage Conditions | -20°C, dry, light-protected |
| Mechanism | Disrupts bacterial membrane function and inhibits peptidoglycan biosynthesis |
| Applications | Antimicrobial research, Gram-positive bacteria studies, antibiotic mechanism exploration |
| Experimental Models | In vitro bacterial cultures, membrane permeability assays, Gram-positive infection models |
| GMP Compliance | Manufactured under GMP conditions |
| Safety Note | For research use only; not for therapeutic or diagnostic use |
| Availability | Wholesale & retail supply |
Friulimicin D’s high purity, GMP-compliant production, and structural consistency ensure reproducibility across antimicrobial assays and mechanistic studies.
Mechanism of Action & Research Applications
Mechanism of Action
Friulimicin D exerts its antibacterial effects through dual mechanisms:
Membrane Interaction:
The lipid tail inserts into bacterial membranes, perturbing membrane integrity and increasing permeability.Cell Wall Biosynthesis Inhibition:
By interfering with peptidoglycan synthesis, Friulimicin D prevents proper cell wall formation, leading to bacterial lysis.
This combination of membrane disruption and enzymatic pathway interference confers potent bactericidal activity, particularly against Gram-positive species such as Staphylococcus aureus, Enterococcus faecalis, and Clostridium species.
Research Applications
Antibiotic Mechanism Studies
Dissecting lipopeptide-target interactions with bacterial membranes
Investigating synergy with ?-lactams, glycopeptides, and other antimicrobials
Multidrug-Resistant Strains Research
Evaluating efficacy against vancomycin-resistant Enterococci (VRE)
Studying mechanisms of resistance development and evasion
In Vitro Assays
MIC (minimum inhibitory concentration) determination
Bacterial growth inhibition curves
Membrane depolarization and permeability assays
In Vivo Preclinical Models
Infection models in murine and rabbit systems
Assessment of lipopeptide pharmacodynamics and pharmacokinetics
Translational Research
Provides a reference compound for novel antibiotic candidates
Contributes to structure–activity relationship (SAR) studies of cyclic lipopeptides

Side Effects (For Reference in Research Models)
Friulimicin D is intended strictly for laboratory research. Experimental observations in bacterial and mammalian model systems include:
Bacterial Adaptation: Dose-dependent emergence of resistant colonies in prolonged exposure studies
Cytotoxicity: Minimal in mammalian cell lines, but high concentrations may affect cell viability
Solvent-Related Effects: DMSO or methanol vehicle effects must be controlled
Stability Considerations: Avoid repeated freeze-thaw cycles to maintain antibacterial activity
Experimental Limitations: Efficacy may vary across bacterial strains and culture conditions
Proper experimental design, including control groups and concentration titration, is essential to ensure valid and reproducible results.
Disclaimer
For laboratory research use only. Not for human or veterinary use.
Keywords
Lipopeptide antibiotic Friulimicin D
Friulimicin D GMP supplier
Actinoplanes friuliensis antibiotic peptide
Gram-positive bacteria research
Friulimicin D antimicrobial mechanism
High purity lipopeptide antibiotic
Friulimicin D preclinical studies
Research use only antibiotic
Novel lipopeptide antibiotic peptide
Additional information
| Weight | 0.8 kg |
|---|---|
| Dimensions | 53 × 42 × 53 cm |














Reviews
There are no reviews yet.