No products in the cart.
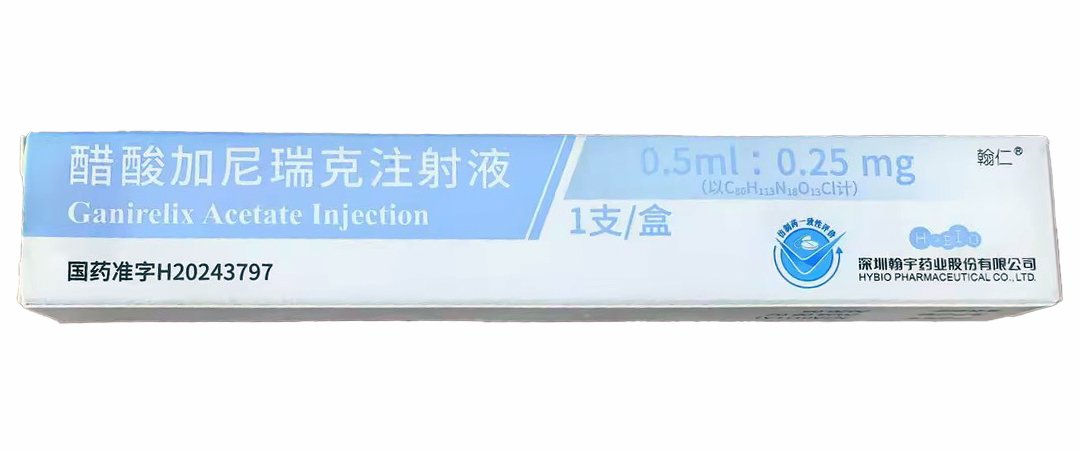
Ganirelix Acetate Injection 0.5?mL:0.25?mg
$2.00
Ganirelix Acetate Injection (0.25?mg per 0.5?mL) is a GnRH antagonist supplied in prefilled syringes by Suzhou Erye Pharmaceutical Co., Ltd. Approved by China NMPA under H20243469, this injection is ideal for fertility and reproductive biology research, including controlled ovarian hyperstimulation models.
?? Strictly for laboratory research use only. Not for therapeutic, clinical, or diagnostic application.?Please consult staff for other specifications and uses?
Description
Ganirelix Acetate Injection Product Specifications
| Parameter | Details |
|---|---|
| Product Name | Ganirelix Acetate Injection |
| Generic Name | Ganirelix Acetate |
| Formulation | Injectable solution (prefilled syringe) |
| Strength | 0.25?mg in 0.5?mL |
| Pack Size | 1 syringe per unit |
| Dosage Form | Subcutaneous injection |
| Approval Number | NMPA China: H20243469 |
| Product Code | 86983233000033 |
| Manufacturer | Suzhou Erye Pharmaceutical Co., Ltd. |
| CAS Number | 124904?93?4 |
| Barcode | 6955872600169 |
| Storage | Store at 20–25?°C (room temp); protect from light |
| Intended Use | Laboratory/fertility research only |
Ganirelix Acetate Injection Mechanism of Action
Ganirelix acetate is a GnRH antagonist that competitively binds GnRH receptors on pituitary gonadotrophs, rapidly suppressing LH and FSH release to prevent premature LH surge during ovarian stimulation fda.report+2accessdata.fda.gov+2en.wikipedia.org+2.
Research Applications & Pharmacology
Controlled ovarian hyperstimulation (COH) models
Fertility and reproductive biology assays
Hormonal regulation and LH/FSH suppression studies
Pharmacokinetics (250?µg dose):
Tmax ~1.1?h, half-life ~16.2?h, 91% bioavailability
Eliminated ~75% in feces, ~22% in urine
Safety Profile & Handling
Common Adverse Reactions (in clinical trials):
Injection site pain, redness, swelling
Abdominal/gastrointestinal pain, nausea, vaginal bleeding, headaches
Rare ovarian hyperstimulation syndrome (OHSS)
Handling & Storage:
Use lab PPE: gloves, mask, coat
For subcutaneous injection only
Store at 20–25?°C, protect from light
Not for human/veterinary use — for lab protocols only
?? Research Use Disclaimer
This product is intended solely for laboratory research use. Not authorized for human, veterinary, clinical, or diagnostic purposes. Misuse may violate regulations and pose health risks.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |









Reviews
There are no reviews yet.