No products in the cart.
Sale
Gimatecan Tablet | CAS 292618-32-7 | High-Purity Topoisomerase I Inhibitor buy low price
Original price was: $3.00.$2.00Current price is: $2.00.
Gimatecan Tablet (CAS 292618-32-7) are high-purity research-grade capsules designed as a potent Topoisomerase I inhibitor. These capsules provide precise dosing for in vitro and preclinical cancer studies, apoptosis research, and molecular oncology applications.
Description
Product Description
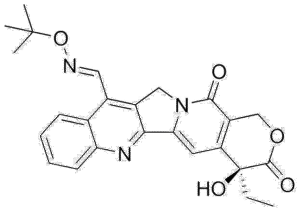
Gimatecan Tablet (CAS 292618-32-7) are advanced research compounds that act as selective Topoisomerase I inhibitors, critical for DNA replication and transcription studies in oncology research. These capsules are designed for laboratory and preclinical use, providing stable, high-purity, and reproducible dosing, which is essential for studying molecular mechanisms of DNA damage, apoptosis, and tumor growth suppression.
Gimatecan functions by binding to the Topoisomerase I-DNA complex, stabilizing transient single-strand breaks, and preventing DNA religation. This mechanism leads to DNA damage accumulation during S-phase, causing replication fork collapse, activation of DNA damage response pathways, and ultimately cell-cycle arrest or apoptosis. Researchers use these capsules to model Topoisomerase I-dependent cytotoxicity in a variety of human cancer cell lines, including colon, lung, and ovarian carcinoma models.
The capsule formulation ensures consistent dosing, minimizes handling errors, and reduces compound degradation compared to powders or solutions. Manufactured under GMP standards, Gimatecan Tablet undergo rigorous quality control, including HPLC, LC-MS/MS, and NMR analysis, confirming identity, purity ≥99%, and stability suitable for long-term preclinical experiments.
Molecular Oncology Applications:
Investigation of DNA damage-mediated apoptosis in cancer cell lines.
Study of replication stress and Topoisomerase I-dependent cytotoxicity.
Preclinical exploration of chemotherapeutic synergy and combination therapy models.
Pathway-specific analysis of p53, ATM/ATR, and Chk1/2 activation.
Assessment of DNA repair mechanisms and molecular targets for drug discovery.
Mechanistic Research Applications:
Quantitative analysis of Topoisomerase I-DNA cleavage complexes.
Measurement of S-phase-dependent DNA strand breaks.
Evaluation of apoptosis and caspase activation in response to Topoisomerase I inhibition.
Assessment of gene expression changes following DNA damage induction.
High-throughput screening of DNA damage response modulators in combination with Gimatecan.
Quality and Consistency:
Gimatecan Tablet are produced with stringent GMP compliance to ensure uniformity and reproducibility. Each batch is validated for purity ≥99%, dissolution profiles, and analytical verification to provide confidence in experimental outcomes.
These capsules allow researchers to perform detailed mechanistic studies on Topoisomerase I inhibition, DNA damage response, and cell-cycle modulation. They are ideal for both academic and industrial laboratories conducting preclinical oncology research, mechanistic pathway analysis, and pharmacodynamic modeling.
By providing a stable, high-purity, and precise capsule format, Gimatecan Tablet are a reliable tool for investigating DNA replication stress, apoptosis induction, and chemotherapeutic mechanisms, supporting the development of novel anti-cancer strategies in preclinical research.
Product Specifications
| Parameter | Specification |
|---|---|
| Chemical Name / Synonyms | Gimatecan, ST1968, Topoisomerase I inhibitor |
| CAS Number | 292618-32-7 |
| Molecular Formula | C₂₆H₃₀N₂O₄ |
| Molecular Weight | 430.5 g/mol |
| Purity | ≥99% (HPLC) |
| Appearance | Light yellow to off-white capsules |
| Dosage Form | Capsules |
| Solubility | Soluble in DMSO; poorly soluble in water; moderate solubility in ethanol |
| Concentration | Available in research-specific strengths for in vitro and preclinical studies |
| Storage Temperature | 2–8°C, protect from moisture and light |
| Stability | Stable for ≥24 months under recommended storage conditions |
| Analytical Methods | HPLC, LC-MS/MS, NMR spectroscopy, FTIR |
| Mechanistic Target | Topoisomerase I |
| Structural Category | Small-molecule DNA replication inhibitor |
| Applications | In vitro DNA damage assays, preclinical oncology research, apoptosis and cell-cycle studies, combinatorial drug evaluation |
| Batch Consistency | GMP-compliant manufacturing ensures uniformity and reproducibility |
| Regulatory Notes | For research use only, not for human or veterinary administration |
| Origin | China, GMP-certified facility |
| Additional Data | Certificates of Analysis (CoA) provided for each batch; stability and dissolution profiles verified |
Detailed Description:
Gimatecan Tablet are formulated to provide high-purity, reproducible dosing for laboratory experiments. Capsules are preferred in preclinical research for precise measurement of Topoisomerase I inhibition effects, minimizing variability and degradation compared to powders or solutions. Each batch is validated to meet strict GMP standards, ensuring consistent molecular activity in experimental setups.
Researchers use these capsules to investigate DNA replication stress, apoptosis pathways, and preclinical anti-cancer mechanisms. By using Gimatecan Capsules, labs can explore combinatorial treatment effects, study pharmacodynamic responses, and map molecular pathways affected by Topoisomerase I inhibition.
The capsule format allows for easy handling, accurate dosing in multi-well plate assays, and compatibility with high-throughput screening, making it an ideal choice for mechanistic oncology studies and pharmacological research.
Mechanism of Action
Gimatecan Tablet (CAS 292618-32-7) function as potent, selective inhibitors of Topoisomerase I, an essential nuclear enzyme that regulates DNA supercoiling during replication and transcription. Topoisomerase I transiently induces single-strand DNA breaks, allowing the relaxation of supercoiled DNA and facilitating the progression of replication forks. Gimatecan stabilizes the covalent Topoisomerase I-DNA cleavage complex, preventing DNA religation and causing accumulation of DNA single-strand breaks. This mechanism leads to replication fork collapse, activation of DNA damage response pathways, and cell-cycle arrest, ultimately inducing apoptosis in proliferating cells.
1. Topoisomerase I Binding and Stabilization
Upon cellular uptake, Gimatecan Tablet interact with the Topoisomerase I-DNA complex, forming a ternary complex that stabilizes single-strand breaks. By preventing the religation step of the Topoisomerase I catalytic cycle, Gimatecan leads to the persistence of DNA nicks. Accumulated DNA damage during S-phase triggers replication fork stalling and collapse, resulting in double-strand breaks detectable in experimental assays. This mechanism makes Gimatecan Tablet highly effective in studying DNA replication stress and related signaling pathways.
2. DNA Damage Response Activation
The accumulation of DNA strand breaks induced by Gimatecan Tablet activates canonical DNA damage response (DDR) pathways, including ATM/ATR, Chk1/Chk2, and p53-mediated checkpoints. Researchers can monitor phosphorylation of H2AX (γH2AX), ATM, and Chk1 as readouts of DDR activation. Activation of these pathways results in:
Cell-cycle arrest: Predominantly in S-phase and G2/M phases.
Apoptosis induction: Upregulation of pro-apoptotic proteins (Bax, Bak) and activation of caspase-3/7.
DNA repair modulation: Engagement of base excision repair (BER) and homologous recombination (HR) pathways.
This mechanistic insight allows researchers to dissect the molecular basis of Topoisomerase I-mediated cytotoxicity and study genetic determinants of response.
3. Apoptosis and Cell-Cycle Effects
Exposure to Gimatecan Tablet results in profound cell-cycle modulation in proliferating cancer cell lines. Key observations include:
S-phase arrest: Direct consequence of replication fork stalling and DNA damage accumulation.
G2/M checkpoint activation: Secondary response to unrepaired DNA lesions.
Apoptotic signaling: Mitochondrial membrane depolarization, caspase cleavage, and PARP fragmentation.
These cellular effects are widely utilized in mechanistic research to explore the relationship between Topoisomerase I inhibition, DNA damage accumulation, and apoptotic responses. The capsules enable reproducible studies of dose-dependent cytotoxicity and mechanism-of-action analysis in various cell models.
4. Molecular Pathway Modulation
Gimatecan Tablet influence multiple downstream signaling pathways through DNA damage induction. Researchers often observe:
Activation of p53 pathway: Transcriptional upregulation of p21, Bax, and GADD45.
Modulation of ATR/Chk1 signaling: Coordination of S-phase checkpoint responses and replication fork stabilization.
Interference with DNA repair machinery: Altered expression of BRCA1/2 and RAD51, providing insights into homologous recombination repair processes.
By evaluating these molecular effects, investigators can study combinatorial drug interactions, synthetic lethality strategies, and predictive biomarkers for Topoisomerase I-targeted therapies.
5. Preclinical and Experimental Applications
The Tablet formulation of Gimatecan allows researchers to:
Perform in vitro cytotoxicity assays using RET-independent and RET-dependent cancer cell lines.
Evaluate replication stress and DNA damage responses quantitatively via γH2AX foci formation and comet assays.
Model drug resistance mechanisms, including alterations in DNA repair pathway activity.
Conduct combinatorial studies with PARP inhibitors, chemotherapeutics, or other kinase inhibitors to explore synergistic effects.
These applications provide a robust platform for mechanistic oncology research, preclinical pharmacology, and molecular pathway validation.
6. Summary
Overall, Gimatecan Capsules act as selective Topoisomerase I inhibitors, stabilizing cleavage complexes, inducing DNA damage, triggering cell-cycle arrest, and activating apoptosis pathways. Their precise, reproducible formulation ensures reliable mechanistic studies, allowing researchers to dissect DNA replication, repair, and apoptosis mechanisms, explore combinatorial therapies, and advance preclinical oncology research.
Applications
Gimatecan Tablet (CAS 292618-32-7) are widely employed in preclinical and molecular oncology research, providing a robust tool for studying Topoisomerase I inhibition, DNA damage responses, apoptosis, and chemotherapeutic mechanisms. The high-purity capsule formulation ensures precise dosing and reproducible results in diverse laboratory applications.
1. Preclinical Cancer Models
Researchers utilize Gimatecan Tablet to explore anticancer effects in vitro and in vivo. Applications include:
Testing cytotoxicity in colon, lung, ovarian, and pancreatic cancer cell lines.
Modeling tumor growth inhibition in xenograft and orthotopic preclinical models.
Investigating dose-response relationships and pharmacodynamic markers such as γH2AX and caspase activation.
Exploring molecular pathways involved in apoptosis, cell-cycle arrest, and DNA repair.
This enables laboratories to validate Topoisomerase I inhibition as a therapeutic strategy and refine preclinical study designs.
2. DNA Damage and Replication Stress Studies
Gimatecan Tablet serve as a tool for examining replication stress and DNA damage mechanisms:
Quantifying single-strand and double-strand DNA breaks using comet assays or γH2AX foci analysis.
Studying S-phase replication fork dynamics and checkpoint activation.
Investigating homologous recombination (HR) and non-homologous end joining (NHEJ) repair pathways.
Dissecting signaling networks involving p53, ATM/ATR, Chk1/Chk2, and downstream effectors.
These studies provide critical insights into the mechanistic basis of Topoisomerase I-targeted cytotoxicity and DNA repair modulation.
3. Apoptosis and Cell-Cycle Research
The capsules are extensively applied in cellular apoptosis and cell-cycle experiments:
Inducing programmed cell death in cancer cell lines for mechanistic studies.
Measuring caspase activation, PARP cleavage, and mitochondrial depolarization.
Evaluating checkpoint-mediated cell-cycle arrest, primarily in S-phase and G2/M phase.
Performing combination studies to enhance apoptosis via synergistic targeting of complementary pathways.
These applications allow researchers to dissect molecular responses to DNA damage and identify potential combinatorial strategies for preclinical therapy.
4. High-Throughput Screening and Drug Discovery
Gimatecan Capsules are suitable for high-throughput screening (HTS) and preclinical drug discovery studies:
Screening novel compounds for synergy or antagonism with Topoisomerase I inhibition.
Evaluating genetic and chemical modulators that influence DNA damage response or apoptotic pathways.
Using multi-well plate formats to test multiple cancer cell lines and treatment conditions.
Standardizing experimental conditions via capsule dosing for reproducible HTS results.
These applications support rapid identification of potential combination therapies and mechanistic validation in laboratory research.
5. Molecular Oncology and Biomarker Studies
Gimatecan Capsules are utilized for molecular pathway and biomarker studies:
Profiling gene expression changes following Topoisomerase I inhibition.
Investigating DNA repair protein expression (e.g., BRCA1, RAD51) under replication stress conditions.
Identifying predictive biomarkers for cytotoxic response and drug sensitivity.
Integrating with transcriptomics and proteomics to map affected molecular networks.
These studies facilitate translational research by connecting molecular mechanisms with potential therapeutic strategies.
6. Summary
In summary, Gimatecan Capsules provide a versatile platform for laboratory and preclinical research, enabling:
Evaluation of anticancer efficacy in cell lines and animal models
Detailed analysis of DNA damage, replication stress, and apoptosis
High-throughput screening and combinatorial drug research
Mechanistic exploration of Topoisomerase I inhibition and molecular signaling
Biomarker discovery and translational research applications
The Tablet formulation ensures reproducible dosing, stability, and high-purity delivery, making Gimatecan Tablet an indispensable tool in modern molecular oncology and pharmacological research.
Side Effects
Gimatecan Tablet (CAS 292618-32-7), while strictly for laboratory and preclinical research, exhibit measurable cellular and molecular effects that are critical for investigators to understand during experimental design. As a potent Topoisomerase I inhibitor, the compound induces DNA damage and replication stress, leading to both intended mechanistic outcomes and observable laboratory effects.
1. Cellular Effects
Exposure to Gimatecan Tablet in cancer cell lines results in cell-cycle arrest, primarily at the S-phase or G2/M transition. Researchers observe a reduction in proliferation markers and alterations in cyclin/CDK activity. Long-term exposure can induce apoptosis, as indicated by caspase-3/7 activation, PARP cleavage, and mitochondrial membrane potential disruption. These effects are dose-dependent and provide reproducible endpoints for mechanistic studies on DNA damage-mediated cytotoxicity.
2. Molecular and Signaling Effects
At the molecular level, Gimatecan stabilizes Topoisomerase I-DNA cleavage complexes, leading to accumulation of single-strand breaks and subsequent replication fork collapse. Key signaling pathways affected include:
ATM/ATR activation: Initiating S-phase checkpoints and DNA repair cascades.
Chk1/Chk2 phosphorylation: Modulating cell-cycle arrest and replication stress responses.
p53 pathway upregulation: Inducing expression of p21, Bax, and GADD45.
These molecular effects allow researchers to study DNA damage responses, repair mechanisms, and apoptotic induction with high specificity.
3. Laboratory Handling Considerations
Despite the capsule formulation reducing handling risks compared to powders, Gimatecan Capsules are bioactive and should be handled with appropriate personal protective equipment (PPE), including gloves, lab coats, and eye protection. Experiments should be conducted within a fume hood or biosafety cabinet to prevent accidental exposure. Proper disposal of residual material and contaminated consumables is essential to maintain laboratory safety and compliance with GLP standards.
4. Off-Target and Experimental Considerations
At higher concentrations, Gimatecan may display off-target effects in certain cell models, especially in genetically altered or overexpressed systems. Researchers should carefully titrate experimental concentrations, validate effects with complementary assays, and employ proper controls to distinguish Topoisomerase I-specific effects from nonspecific cytotoxicity.
5. Summary
In preclinical research, the observed side effects of Gimatecan Tablet—including S-phase arrest, apoptosis induction, and activation of DNA damage signaling pathways—are integral to their mechanism of action. Understanding these effects allows researchers to design robust and reproducible experiments while ensuring laboratory safety.
These characteristics make Gimatecan Tablet a valuable tool for molecular oncology research, DNA damage studies, and mechanistic pharmacology experiments.
Keywords
Gimatecan Tablet, CAS 292618-32-7, Topoisomerase I inhibitor, DNA replication inhibitor, anticancer research compound, preclinical oncology reagent, apoptosis research tool, cell-cycle study reagent, replication stress modulator, molecular oncology research chemical, high-purity small molecule, bulk production research-grade compound, GMP-grade research chemical, factory small-molecule supplier, China chemical manufacturer, combinatorial therapy screening reagent, DNA damage response assay, high-throughput screening tool.
These keywords are optimized for visibility in scientific catalogs, research procurement platforms, and academic databases, ensuring maximum discoverability for laboratories seeking high-purity Gimatecan Tablet for preclinical studies and mechanistic experiments.
Shipping Guarantee
Gimatecan Tablet (CAS 292618-32-7) are shipped under strict temperature-controlled conditions to preserve compound integrity, stability, and bioactivity. Cold-chain logistics, insulated packaging, and tamper-evident containers minimize degradation during transport.
Real-time tracking and insurance coverage are included to ensure delivery integrity. Special arrangements for international shipping, express delivery, and temperature-sensitive handling are available to guarantee capsules arrive in optimal condition for research purposes.
Trade Assurance
Every batch of Gimatecan Tablet undergoes GMP-compliant manufacturing and quality control verification. Purity ≥99% is confirmed via HPLC, LC-MS/MS, and NMR analysis, and a Certificate of Analysis (CoA) is provided for each lot.
Bulk production, OEM, and customized capsule strengths are available to meet the needs of academic, industrial, and preclinical research laboratories. A reliable return or replacement policy ensures consistent, research-grade compound quality for experimental reproducibility.
Payment Support
We support secure and flexible payment options, including PayPal, major credit cards, T/T bank transfers, USDT, Bitcoin, and Ethereum. All transactions are fully encrypted to protect sensitive financial information.
This allows global research institutions, preclinical labs, and industrial R&D centers to acquire high-purity Gimatecan Tablet efficiently, facilitating uninterrupted experimental workflows and large-scale procurement for mechanistic and preclinical studies.
Disclaimer
Gimatecan Tablet (CAS 292618-32-7) are intended strictly for laboratory and preclinical research use. They are not for human or veterinary administration. Researchers must follow institutional biosafety guidelines, use appropriate PPE, and handle the compound according to GLP standards.
Improper use may alter DNA damage responses and experimental outcomes. The information provided herein is solely for scientific research and educational applications, and users are responsible for safe storage, handling, and disposal of all materials.
References
PubChem – Gimatecan
Provides chemical structure, molecular formula, physicochemical properties, and bioactivity data of Gimatecan Capsules for laboratory research and preclinical studies.ChEMBL – Gimatecan
Contains detailed bioactivity information, molecular target interactions, and mechanistic data suitable for Topoisomerase I inhibition research.DrugBank – Gimatecan
Provides pharmacological profiles, mechanism of action, and preclinical research applications of Gimatecan Capsules.PubMed – Gimatecan Research Articles
Access peer-reviewed publications detailing Topoisomerase I inhibition, DNA damage response, and preclinical oncology studies using Gimatecan.IUPHAR/BPS – Kinase and DNA Damage Targets
Provides curated data on molecular targets, DNA damage pathways, and signaling mechanisms relevant to Gimatecan Capsules research.Additional Resources
ACS Chemical Biology Journal Articles on Topoisomerase I Inhibitors.
Sigma-Aldrich Technical Datasheets.
ResearchGate Experimental Studies on Gimatecan.
These references offer authoritative, peer-reviewed information for laboratories using Gimatecan Capsules (CAS 292618-32-7) in mechanistic, preclinical, and translational research applications. Researchers can access molecular data, pharmacology, and experimental protocols to design reproducible, high-impact studies in oncology, apoptosis, and DNA damage response research.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
1. What is the primary research use of Gimatecan Tablet?
Gimatecan Tablet (CAS 292618-32-7) are primarily used as a Topoisomerase I inhibitor for preclinical oncology research. They enable studies on DNA replication stress, apoptosis induction, and mechanistic pathway analysis.
2. How should Gimatecan Tablet be stored?
Store the Tablet at 2–8°C, away from light and moisture. Proper storage ensures stability, reproducibility, and high-purity performance during laboratory experiments.
3. Are Gimatecan Tablet GMP-compliant?
Yes, all Tablet are manufactured under GMP standards, ensuring consistent batch-to-batch purity ≥99%, identity verification, and reliability for mechanistic research studies.
4. Can Gimatecan Tablet be used in combination therapy studies?
Yes, researchers often use Gimatecan Tablet with other chemotherapeutic agents or molecular inhibitors to investigate synergistic or additive effects on DNA damage and apoptosis pathways.
5. Is a Certificate of Analysis (CoA) provided?
Every batch of Gimatecan Tablet includes a CoA confirming purity, identity, and analytical testing via HPLC, LC-MS/MS, and NMR, supporting reproducible experimental results.
6. Can bulk production or OEM services be requested?
Yes, our factory small-molecule supplier provides bulk production, OEM services, and customized capsule strengths to meet the needs of academic and industrial laboratories.
7. Are Gimatecan Tablet suitable for apoptosis research?
Yes, Gimatecan Tablet induce replication-dependent apoptosis in cancer cell lines, enabling the study of caspase activation, mitochondrial signaling, and DNA damage-mediated cell death.
8. Can these Tablet be used for high-throughput screening assays?
Absolutely. Gimatecan Tablet are compatible with multi-well plate formats for screening DNA damage response modulators and potential combination therapies in preclinical studies.
9. How do Gimatecan Tablet affect DNA replication?
By stabilizing the Topoisomerase I-DNA cleavage complex, these Tablet cause replication fork stalling, DNA damage accumulation, and activation of cell-cycle checkpoints, enabling mechanistic studies of replication stress.
10. What cell lines are typically used with Gimatecan Tablet?
Researchers commonly use colon, lung, ovarian, and pancreatic cancer cell lines, but the Tablet are applicable in any proliferative cell model for mechanistic and pharmacological investigations.
11. What safety precautions should be taken in the lab?
Always use PPE and work in a fume hood or biosafety cabinet. Proper containment, handling, and disposal are essential to maintain lab safety while using Gimatecan Tablet.
12. Can Gimatecan Tablet be used in pharmacokinetic studies?
Yes, they are suitable for preclinical PK/PD studies, allowing researchers to assess systemic exposure, target engagement, and Topoisomerase I inhibition in vivo models.
13. Are the Tablet stable for long-term studies?
Yes, Gimatecan Tablet remain stable for ≥24 months under recommended storage conditions, ensuring consistent experimental results for extended preclinical research.
14. How can researchers confirm Tablet dosage accuracy?
The capsule formulation ensures precise dosing, reducing variability in in vitro and preclinical studies and enabling reliable concentration-dependent experiments.
15. Who supplies high-purity Gimatecan Tablet globally?
Gimatecan Tablet are supplied by reputable China chemical manufacturers and factory small-molecule suppliers offering high-purity bulk production suitable for preclinical research, mechanistic studies, and combinatorial screening assays.
















Reviews
There are no reviews yet.