No products in the cart.
Sale
Nintedanib Esylate CAS 656247-18-6 – Factory Manufactured, Low Price Wholesale | Buy Online
Original price was: $280.00.$260.00Current price is: $260.00.
High-purity Nintedanib Esylate (CAS 656247‑18‑6), a potent multi-target tyrosine kinase inhibitor for research use. Ideal for anti-fibrotic, anti-angiogenic, and oncology studies. Bulk supply and ready-to-use solution ensure reproducibility in in vitro and in vivo models.
Description
Contents
hide
Product Description
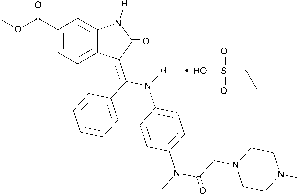
Nintedanib Esylate (CAS 656247‑18‑6) is a high-purity, factory-manufactured small-molecule tyrosine kinase inhibitor (TKI) designed for advanced research in oncology, fibrosis, and vascular biology. This compound selectively inhibits multiple receptor tyrosine kinases, including VEGFR, PDGFR, and FGFR, enabling detailed mechanistic studies of angiogenesis, cellular proliferation, and extracellular matrix remodeling. Provided as a ready-to-use solution, Nintedanib Esylate ensures consistent dosing, high reproducibility, and convenience for both in vitro and in vivo experiments.
In in vitro studies, Nintedanib Esylate is widely applied to investigate VEGFR-driven angiogenesis, PDGFR-mediated fibroblast activation, and FGFR-dependent proliferation in cancer cell lines, endothelial cells, and fibroblasts. Its solution format facilitates dose-response assays, high-throughput screening, and combination therapy experiments with other anti-proliferative or anti-fibrotic compounds. Additionally, it supports organoid and 3D tissue models, allowing exploration of tumor microenvironments and fibrotic tissue remodeling mechanisms.
In in vivo research, Nintedanib Esylate is extensively used in rodent models of cancer, pulmonary fibrosis, and vascular dysfunction. Researchers can evaluate tumor growth inhibition, angiogenesis suppression, extracellular matrix modulation, and systemic pharmacodynamic responses. Its broad-spectrum kinase inhibition allows detailed analysis of receptor crosstalk and compensatory signaling pathways, enhancing translational research and combination therapy studies. The solution form simplifies accurate administration through intravenous, intraperitoneal, or oral routes, improving experimental reliability.
Manufactured under strict quality control, Nintedanib Esylate achieves ≥ 98% purity confirmed by HPLC, with Certificates of Analysis (COA) and LC–MS documentation provided for each batch. Its stability and high bioactivity make it an ideal tool for integrating multi-omic analyses such as transcriptomics, proteomics, and metabolomics. Bulk supply, ready-to-use formulation, and cost-effective pricing support large-scale preclinical studies, high-throughput screening, and advanced translational research applications in oncology, fibrosis, and vascular biology.
Product Specifications
| Specification | Details |
|---|---|
| Chemical Name | Nintedanib Esylate |
| CAS Number | 656247‑18‑6 |
| Molecular Formula | C₃₁H₃₄N₄O₄·C₂H₆O₃S |
| Molecular Weight | 539.67 g/mol |
| Appearance | Yellow crystalline powder / Clear solution |
| Purity | ≥ 98% (HPLC verified) |
| Solubility | Soluble in DMSO, ethanol; stable in aqueous buffers |
| Storage Conditions | 2–8°C, protected from light |
| Batch Consistency | Factory-controlled; COA provided for each lot |
| Application | In vitro, in vivo, and translational research in oncology, fibrosis, and vascular biology |
| Form | Ready-to-use solution for precise dosing |
| Stability | Stable for 12 months under recommended conditions |
Notes:
Ensures accurate dosing across experimental systems, minimizing variability.
Supports high-throughput screening and reproducible preclinical studies.
Compatible with combination therapy assays, organoid cultures, and 3D tissue models.
Each batch undergoes rigorous quality control to maintain consistent purity and bioactivity.
Ideal for integrating multi-omic analyses and mechanistic studies in angiogenesis, fibrosis, and cancer research.
Mechanism of Action
Nintedanib Esylate (CAS 656247‑18‑6) functions as a potent multi-target tyrosine kinase inhibitor, blocking VEGFR1-3, PDGFRα/β, and FGFR1-3 signaling pathways. By inhibiting these kinases, Nintedanib prevents phosphorylation of downstream proteins involved in angiogenesis, fibroblast proliferation, and extracellular matrix deposition. This mechanism underlies its anti-fibrotic, anti-angiogenic, and anti-proliferative effects observed in experimental models.
In oncology research, VEGFR inhibition disrupts tumor angiogenesis, reducing blood supply and limiting tumor growth. PDGFR blockade interferes with fibroblast activation and pericyte recruitment, while FGFR inhibition modulates cell proliferation and survival pathways. These coordinated effects allow researchers to study complex signaling crosstalk and compensatory mechanisms in cancer and fibrotic tissue models.
At the cellular level, Nintedanib Esylate prevents tyrosine phosphorylation, modulating receptor-mediated downstream cascades such as MAPK/ERK and PI3K/AKT pathways. This regulation affects endothelial cell migration, tube formation, and fibroblast differentiation. Its multi-target profile also allows combination studies with chemotherapeutics or anti-fibrotic agents to evaluate synergistic or additive effects.
The solution form ensures precise dosing and uniform exposure in both in vitro and in vivo studies, improving reproducibility of pharmacodynamic and mechanistic data. Nintedanib Esylate’s activity is dose-dependent, emphasizing the importance of careful titration and time-course studies to dissect its effects on receptor signaling networks.
Overall, Nintedanib Esylate (CAS 656247‑18‑6) provides a robust tool for investigating receptor tyrosine kinase biology, anti-fibrotic mechanisms, and anti-angiogenic pathways in preclinical research, translational studies, and multi-omic experimental workflows.
Applications
Nintedanib Esylate (CAS 656247‑18‑6) is widely applied in oncology, fibrosis, and vascular biology research. In cancer studies, it is used to inhibit VEGFR, PDGFR, and FGFR signaling, reducing tumor angiogenesis, cell proliferation, and metastasis potential. Researchers can model tumor microenvironment interactions, study anti-angiogenic therapies, and assess combination strategies with chemotherapeutic agents.
In fibrosis research, Nintedanib Esylate modulates fibroblast activation, extracellular matrix deposition, and tissue remodeling. It is particularly useful in pulmonary fibrosis models, enabling investigation of anti-fibrotic pathways, cytokine regulation, and receptor-mediated signaling. Its activity supports translational studies of novel therapeutics and mechanistic exploration of fibrotic disease progression.
The compound also serves in vascular biology research, facilitating the study of endothelial cell function, tube formation, and angiogenic signaling pathways. Its multi-target kinase inhibition allows researchers to dissect complex signaling crosstalk in endothelial and perivascular cells, providing insights into vascular remodeling and pathophysiology.
Nintedanib Esylate is compatible with in vitro, in vivo, and organoid models, enabling reproducible results across experimental systems. Its solution format ensures precise dosing and stable exposure, essential for high-throughput screening, combination therapy experiments, and mechanistic studies. Overall, Nintedanib Esylate provides a versatile platform for exploring anti-angiogenic, anti-fibrotic, and anti-proliferative mechanisms in translational preclinical research.
Research Models
Nintedanib Esylate (CAS 656247‑18‑6) is a versatile multi-target tyrosine kinase inhibitor (TKI) widely employed across a variety of preclinical research models, enabling detailed investigation of angiogenesis, fibrosis, and tumor biology. In oncology research, human cancer cell lines such as lung, breast, and colon carcinoma are commonly used to evaluate anti-proliferative and anti-angiogenic effects. These in vitro models allow precise dose-response studies, mechanistic exploration of VEGFR, PDGFR, and FGFR inhibition, and the assessment of combination therapy strategies with chemotherapeutic agents or other targeted inhibitors. Researchers often utilize 3D spheroid cultures and organoids derived from tumor cells to more accurately recapitulate the tumor microenvironment, enabling studies of vascularization, tumor-stroma interactions, and drug penetration.
In fibrosis research, Nintedanib Esylate is applied to both primary fibroblast cultures and tissue-specific models, including lung, liver, and kidney fibrosis. Rodent models, such as bleomycin-induced pulmonary fibrosis, provide in vivo platforms to study extracellular matrix deposition, fibroblast proliferation, and tissue remodeling under controlled experimental conditions. The compound’s inhibition of PDGFR and FGFR pathways allows researchers to dissect the signaling networks involved in fibroblast activation and collagen deposition, offering insights into anti-fibrotic mechanisms that are highly relevant for translational studies.
For vascular biology research, endothelial cell cultures, angiogenesis assays, and in vivo vessel formation models are widely utilized. These systems help investigate VEGFR-mediated endothelial proliferation, migration, and tube formation, allowing assessment of anti-angiogenic properties. Nintedanib Esylate’s multi-target inhibition profile makes it particularly valuable for studying receptor crosstalk, compensatory signaling, and vascular remodeling in both healthy and pathological tissues.
Integration with multi-omic analyses is feasible across all research models. Researchers can combine transcriptomic, proteomic, phosphoproteomic, and metabolomic approaches to evaluate molecular responses to nintedanib esylate treatment. Rodent and organoid models support high-throughput screening, combination therapy evaluation, and mechanistic studies of disease progression. Moreover, the solution form ensures precise dosing and reproducibility across experiments, enhancing the reliability of data obtained from diverse preclinical systems.
Overall, Nintedanib Esylate (CAS 656247‑18‑6) provides a comprehensive and adaptable platform for investigating kinase-mediated pathways in oncology, fibrosis, and vascular biology, supporting translational and mechanistic studies that bridge in vitro findings with in vivo models and multi-omic analyses.
Experimental Design Considerations
When designing experiments with Nintedanib Esylate (CAS 656247‑18‑6), researchers should consider dose optimization, treatment duration, and model selection. Dose-response studies are essential to determine effective concentrations that inhibit target kinases without inducing off-target cytotoxicity. Time-course studies help capture dynamic receptor signaling and downstream effects.
Choice of research model impacts experimental outcomes. In vitro studies allow precise control over exposure and mechanistic interrogation of VEGFR, PDGFR, and FGFR pathways. In vivo models provide translational insights into tumor angiogenesis, fibrosis progression, and vascular remodeling. Organoid or 3D culture systems enable complex tissue-level studies and drug interaction assessments.
Proper controls are critical, including vehicle-treated and untreated samples, to distinguish compound-specific effects from background variability. For combination therapy studies, sequential or simultaneous dosing schedules should be carefully designed to evaluate synergistic, additive, or antagonistic effects.
Preparation and handling of Nintedanib Esylate solution must ensure accurate concentration and sterility, especially for sensitive assays. Researchers should document all experimental parameters, including batch number, storage conditions, and administration routes, to maintain reproducibility. Integration with multi-omic and computational analysis enhances mechanistic understanding and supports robust translational study design.
Laboratory Safety & Handling Guidelines
Nintedanib Esylate (CAS 656247‑18‑6) should be handled under standard laboratory safety protocols. Use personal protective equipment (PPE), including gloves, lab coat, and safety goggles. Work in a well-ventilated area or chemical fume hood to minimize exposure to dust or aerosols.
Store the compound at 2–8°C, protected from light, and avoid repeated freeze-thaw cycles. Use sterile pipettes or syringes to maintain solution integrity. Handle with care to avoid skin contact, inhalation, or ingestion. Follow institutional procedures for hazardous chemical waste disposal.
When preparing experimental solutions, use precise volumetric techniques to ensure reproducible dosing. Label all vials clearly with concentration, date, and batch number. Maintain an accessible Safety Data Sheet (SDS) for emergency reference, including spill management and first aid instructions.
For combination or high-throughput studies, consider cumulative exposure and potential interactions with other reagents. Proper documentation and adherence to handling guidelines help maintain compound stability, ensure reliable experimental results, and protect personnel in preclinical research settings.
Integration with Multi‑Omic & Computational Studies
Nintedanib Esylate (CAS 656247‑18‑6) integrates seamlessly into multi-omic research workflows. Inhibition of VEGFR, PDGFR, and FGFR pathways can be monitored across transcriptomic, proteomic, phosphoproteomic, and metabolomic platforms, allowing systems-level analysis of signaling cascades.
The compound supports computational modeling, including receptor-ligand interactions, pathway simulations, and network analyses. Combining in vitro or in vivo experimental data with bioinformatics pipelines enables correlation of multi-omic datasets with physiological outcomes, providing mechanistic insights into anti-angiogenic and anti-fibrotic effects.
Researchers can use Nintedanib Esylate to identify novel biomarkers, elucidate crosstalk between signaling networks, and predict therapeutic responses in translational models, enhancing the utility of both experimental and computational studies.
Keywords
nintedanib esylate, CAS 656247‑18‑6, tyrosine kinase inhibitor, TKI, multi-target kinase inhibitor, anti-fibrotic, anti-angiogenic, VEGFR inhibitor, PDGFR inhibitor, FGFR inhibitor, oncology research, fibrosis research, vascular biology, in vitro, in vivo, translational research
Shipping Guarantee
Nintedanib Esylate (CAS 656247‑18‑6) is shipped globally using express carriers with real-time tracking to ensure timely and reliable delivery to research laboratories. Each package is carefully prepared with temperature-controlled packaging to preserve compound stability during transit, minimizing degradation or loss of bioactivity. Moisture-resistant sealing further protects the solution from environmental fluctuations, ensuring consistent experimental performance upon arrival. Every shipment includes a batch-specific Certificate of Analysis (COA), allowing researchers to verify purity, potency, and lot consistency before beginning experiments. The combination of controlled packaging, tracking, and documentation provides researchers with confidence in the integrity of the compound, whether for in vitro assays, in vivo studies, or integration into multi-omic research workflows. Nintedanib Esylate’s shipping procedures are designed to support reproducible and reliable preclinical studies while maintaining the highest standards of compound handling throughout the supply chain.
Trade Assurance
Our trade assurance program for Nintedanib Esylate ensures that bulk orders are supported with verified documentation, including Certificates of Analysis (COA), HPLC, and LC–MS reports for each batch. Factory-controlled production processes maintain batch-to-batch consistency, providing reliable compound quality for large-scale preclinical and translational research. Institutional clients can access secure commercial contracts, facilitating procurement for high-throughput screening, long-term studies, and combination therapy experiments. The trade assurance framework also guarantees proper handling, packaging, and documentation during manufacturing and distribution, minimizing the risk of errors or variability. Researchers benefit from predictable supply and quality control, enabling reproducible experimental results across multiple labs and studies. This structured approach reinforces confidence in Nintedanib Esylate as a dependable multi-target tyrosine kinase inhibitor for oncology, fibrosis, and vascular biology research.
Payment Support
Flexible payment options are available for Nintedanib Esylate to accommodate the diverse needs of research laboratories. Accepted methods include bank transfer, PayPal, telegraphic transfer (TT), letters of credit (LC), and corporate invoicing, providing convenience for domestic and international clients. For small-scale experimental use, researchers can order sample quantities with simplified payment procedures, while bulk orders for institutional studies are supported with detailed commercial documentation. These options enable laboratories to manage budgets efficiently, plan long-term research projects, and ensure timely procurement. Payment processes are secure, compliant with international financial regulations, and designed to facilitate smooth transactions for preclinical studies. By offering versatile and reliable payment support, researchers can focus on experimental design, dose optimization, and mechanistic studies using Nintedanib Esylate, without concerns about procurement or financial logistics.
Disclaimer
Nintedanib Esylate (CAS 656247‑18‑6) is intended strictly for research purposes only and is not approved for human or veterinary use. It must be handled by trained personnel in accordance with institutional safety protocols, including appropriate use of personal protective equipment (PPE), fume hoods, and controlled laboratory environments. The compound should not be ingested, injected, or administered outside of experimental research settings. All experiments must adhere to ethical and legal regulations relevant to preclinical studies. Researchers are responsible for proper storage, handling, and disposal of Nintedanib Esylate to prevent exposure, contamination, or environmental impact. The manufacturer and supplier disclaim any liability for unintended or improper use. Observed effects, side effects, and experimental outcomes described in preclinical studies reflect controlled laboratory conditions and do not translate to clinical or therapeutic applications. This disclaimer ensures safe and responsible use of the compound while supporting reproducible scientific research.
References
Boehringer Ingelheim — Nintedanib Esylate technical and pharmacological profile.
Cayman Chemical — Tyrosine kinase inhibitor product datasheet.
Selleck Chemicals — Nintedanib HCl/Esylate bioactivity and preclinical applications.
Tocris / Bio-Techne — Multi-target kinase inhibitor research information.
PubChem — Chemical structure and properties for Nintedanib Esylate (CAS 656247‑18‑6).

Additional information
| Weight | 0.8 kg |
|---|---|
| Dimensions | 56 × 42 × 56 cm |
1 review for Nintedanib Esylate CAS 656247-18-6 – Factory Manufactured, Low Price Wholesale | Buy Online
1. What is Nintedanib Esylate used for in research?
Nintedanib Esylate (CAS 656247‑18‑6) is a multi-target tyrosine kinase inhibitor used to study angiogenesis, fibrosis, and tumor proliferation. It is widely applied in oncology, pulmonary fibrosis, and vascular biology research.
2. Can Nintedanib Esylate be used for clinical treatment?
No, Nintedanib Esylate is strictly for laboratory research. It is not approved for human or veterinary use and should be handled only by trained personnel.
3. Which models are compatible with Nintedanib Esylate research?
It can be used in cell lines, organoids, 3D tissue models, and rodent in vivo studies. These models allow investigation of VEGFR, PDGFR, and FGFR signaling pathways.
4. How should Nintedanib Esylate be stored?
Store at 2–8°C, protected from light, and avoid repeated freeze-thaw cycles. Proper storage ensures stability and consistent experimental results.
5. What are typical concentration ranges for experiments?
Concentrations vary depending on the model, typically ranging from nanomolar to low micromolar. Dose-response studies are recommended to optimize effects.
6. Can it be used in combination studies?
Yes, Nintedanib Esylate can be combined with chemotherapeutic agents or other kinase inhibitors. Experimental controls are essential to assess potential synergistic or additive effects.
7. Is Nintedanib Esylate suitable for high-throughput screening?
Yes, its ready-to-use solution format supports reproducible dosing in high-throughput assays. It is compatible with both in vitro and in vivo screening workflows.
8. How should Nintedanib Esylate be handled in the lab?
Always use PPE, work in a fume hood if possible, and avoid ingestion or direct contact. Follow institutional safety guidelines for hazardous chemicals.
9. What are observed side effects in research models?
Common effects include inhibition of endothelial cell proliferation, fibroblast activation, and tumor growth. Minimal off-target toxicity occurs at recommended experimental concentrations.
10. Does Nintedanib Esylate support multi-omic studies?
Yes, it can be integrated with transcriptomics, proteomics, phosphoproteomics, and metabolomics. This enables comprehensive analysis of molecular pathways.
11. How is the solution prepared for experiments?
Use sterile pipettes and accurate volumetric techniques to ensure reproducible dosing. Proper preparation avoids variability and maintains bioactivity.
12. Are Certificates of Analysis provided?
Yes, each batch comes with COA, HPLC, and LC–MS documentation. This ensures reproducibility and validates compound purity and potency.
13. How long is Nintedanib Esylate stable?
Stable for 12 months under recommended storage conditions. Avoid exposure to extreme temperatures or light to preserve compound integrity.
14. What is the appearance of the compound?
Nintedanib Esylate appears as a yellow crystalline powder or a clear ready-to-use solution. Its appearance confirms correct formulation for research applications.
15. How is shipping handled for research orders?
Global express shipping with tracking ensures timely delivery. Temperature-controlled and moisture-resistant packaging maintains compound integrity during transit.














kaaba –
Paketet är borttappat, skicka mig ett nytt, tack