No products in the cart.
Sale
Ribociclib Succinate Tablets CAS 1374639-75-4 | High-purity bulk wholesale
Original price was: $3.00.$2.00Current price is: $2.00.
Ribociclib Succinate Tablets (CAS 1374639-75-4) are high-purity CDK4/6 inhibitors supplied in a stable oral tablet formulation. Manufactured under GMP-compliant factory conditions, these tablets ensure consistent dosing for preclinical cancer research, including studies on breast cancer, hematologic malignancies, and cell cycle modulation. Wholesale supply and online ordering options are available for bulk research use.
Description
Product Description
1. Molecular Characteristics and Formulation
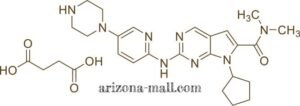
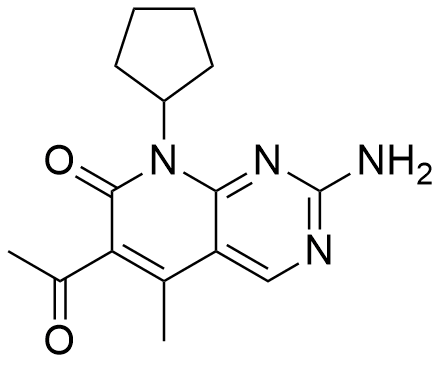
Ribociclib Succinate Tablets are formulated as orally bioavailable tablets containing Ribociclib succinate, a selective cyclin-dependent kinase 4/6 (CDK4/6) inhibitor. By targeting CDK4 and CDK6, Ribociclib prevents phosphorylation of the retinoblastoma protein (Rb), resulting in G1-phase cell cycle arrest. The tablets are high-purity, stable, and manufactured under GMP-compliant conditions, ensuring reproducibility for in vitro and in vivo preclinical studies.
2. Applications in Oncology Research
Ribociclib Succinate Tablets are widely applied in preclinical oncology research, especially in ER-positive/HER2-negative breast cancer models, to investigate tumor proliferation, cell cycle regulation, and combination therapy strategies. By selectively inhibiting CDK4/6 activity, the tablets reduce Rb phosphorylation, halt G1-to-S phase progression, and suppress tumor cell growth. Researchers utilize Ribociclib for monotherapy studies, as well as synergistic combinations with endocrine therapies, targeted inhibitors, or novel anticancer agents.
3. Mechanistic Applications in Cell Cycle Studies
The tablets provide a precise tool for studying the CDK4/6-Rb-E2F pathway. In cell line experiments, Ribociclib induces G1-phase arrest, downregulates E2F target genes, and reduces DNA synthesis, allowing detailed analysis of cell cycle checkpoints, proliferation dynamics, and anticancer drug mechanisms.
4. Pharmacological and Translational Research
As a well-characterized CDK4/6 inhibitor, Ribociclib Tablets are used to evaluate pharmacokinetics (PK), pharmacodynamics (PD), dose-response relationships, and therapeutic efficacy in preclinical models. They enable biosimilar testing, combination therapy studies, and mechanistic investigations, bridging preclinical experiments to potential clinical translation.
Product Specifications
| Parameter | Specification / Details |
|---|---|
| Product Name | Ribociclib Succinate Tablets |
| CAS Number | 1374639-75-4 |
| Molecular Formula | C₂₇H₃₁N₇O•C₄H₆O₄ (succinate salt) |
| Molecular Weight | 434.6 g/mol (base), 504.5 g/mol (succinate salt) |
| Product Type | Selective CDK4/6 inhibitor, orally bioavailable tablets |
| Formulation | High-purity succinate salt in compressed tablets |
| Appearance | White to off-white solid tablets |
| Purity | ≥ 99% (HPLC validated) |
| Dosage Strengths | 50 mg, 100 mg, 200 mg tablets (customizable) |
| Solubility | Slightly soluble in water, soluble in DMSO |
| pH | N/A (solid formulation) |
| Stability | 24 months under recommended storage |
| Storage Conditions | Store in cool, dry, dark place (2–8°C recommended) |
| Packaging Options | Blister packs, bottles, bulk supply for research institutions |
| Customization | Private label and concentration customization available |
| Documentation | COA, MSDS, TDS provided with each lot |
Mechanism of Action
1. CDK4/6 Inhibition
Ribociclib Succinate is a selective inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6). These kinases phosphorylate the retinoblastoma protein (Rb), a key regulator of G1-to-S phase transition. By inhibiting CDK4/6 activity, Ribociclib prevents Rb phosphorylation, maintaining it in its active, hypophosphorylated state, which suppresses E2F transcription factor activation and halts cell cycle progression.
2. Induction of G1-Phase Cell Cycle Arrest
Through CDK4/6 inhibition, Ribociclib Tablets induce cell cycle arrest in the G1 phase. This prevents cells from entering the S phase, thereby reducing DNA replication and proliferation. Experimental models consistently show that Ribociclib decreases cyclin E and E2F target gene expression, confirming effective cell cycle blockade.
3. Tumor Growth Suppression
By arresting the cell cycle, Ribociclib suppresses tumor cell proliferation and limits tumor mass expansion in preclinical studies. Its effects are particularly pronounced in ER-positive/HER2-negative breast cancer models, where CDK4/6-Rb-E2F signaling is a critical driver of tumor progression. Researchers can quantify tumor cell viability, proliferation markers (Ki-67), and Rb phosphorylation status to assess Ribociclib efficacy.
4. Combination Therapy Potential
Ribociclib is commonly used in combination with endocrine therapy (e.g., letrozole, fulvestrant) or targeted inhibitors to enhance therapeutic efficacy. By synchronizing cells in G1, Ribociclib can sensitize tumor cells to additional treatments, providing a robust preclinical tool to study synergistic antitumor effects and resistance mechanisms.
5. Immunomodulatory and Off-Target Effects
While Ribociclib primarily targets CDK4/6, studies indicate potential off-target modulation of cell cycle-related pathways in immune and stromal cells. This property enables investigation of tumor microenvironment interactions, immune checkpoint modulation, and cytotoxic responses in co-culture and organoid models.
6. Translational Research Applications
The well-characterized mechanism of Ribociclib Tablets makes them ideal for pharmacokinetic (PK) and pharmacodynamic (PD) studies, dose-response evaluations, and preclinical efficacy testing. Researchers can combine in vitro mechanistic studies with in vivo xenograft and PDX models to explore therapeutic potential, drug resistance, and biomarker discovery.
Applications
1. Preclinical Oncology Research
Ribociclib Succinate Tablets are widely utilized in preclinical cancer research to study tumor cell proliferation, cell cycle dynamics, and therapeutic responses. Their selective inhibition of CDK4/6 enables precise control over G1-to-S phase transition, making them essential for evaluating anticancer strategies in ER-positive/HER2-negative breast cancer models, prostate cancer, and other solid tumors. Researchers can measure cell proliferation markers, Rb phosphorylation, and DNA synthesis rates to assess efficacy.
2. Hematologic Malignancy Models
Ribociclib Tablets are applied in leukemia and lymphoma studies, where dysregulated CDK4/6-Rb signaling drives uncontrolled proliferation. Preclinical models demonstrate that G1-phase arrest induced by Ribociclib reduces tumor cell viability, facilitates apoptosis induction, and can enhance sensitivity to standard chemotherapeutics. Flow cytometry and molecular assays are commonly used to monitor cell cycle distribution, apoptosis, and signaling pathway modulation.
3. Combination Therapy Research
Ribociclib is extensively studied in combination with endocrine therapies (e.g., letrozole, fulvestrant) or targeted inhibitors (e.g., PI3K/mTOR pathway inhibitors) to evaluate synergistic antitumor effects. Preclinical studies indicate enhanced tumor growth suppression, improved cell cycle control, and delayed resistance development. These applications provide insights into optimized treatment strategies for both solid and hematologic tumors.
4. Cell Cycle Mechanistic Studies
Researchers leverage Ribociclib Tablets to dissect the CDK4/6-Rb-E2F axis, allowing detailed study of cell cycle checkpoints, proliferation kinetics, and oncogenic signaling pathways. In vitro and ex vivo assays, including BrdU incorporation, EdU labeling, and flow cytometry, are used to quantify DNA synthesis inhibition and G1-phase accumulation, providing mechanistic understanding of tumor biology.
5. Drug Resistance and Sensitivity Studies
Ribociclib serves as a tool to investigate resistance mechanisms associated with CDK4/6 inhibition. Preclinical studies focus on Rb loss, cyclin E overexpression, and alternative pathway activation. By combining Ribociclib with other chemotherapeutic or targeted agents, researchers can evaluate strategies to overcome resistance, optimize dose regimens, and improve therapeutic efficacy.
6. Translational and Pharmacology Research
As a well-characterized CDK4/6 inhibitor, Ribociclib Tablets are used in pharmacokinetic (PK), pharmacodynamic (PD), and dose-response studies. These studies inform preclinical modeling, including xenograft, PDX, and organoid experiments, providing critical data for biomarker identification, therapeutic index assessment, and translational oncology research.
7. Immuno-Oncology and Tumor Microenvironment Studies
Ribociclib’s cell cycle modulation indirectly affects the tumor immune microenvironment, influencing immune cell infiltration, cytokine expression, and stromal cell interactions. Preclinical research utilizes Ribociclib to explore immunomodulatory effects, including enhancement of checkpoint inhibitor efficacy or modulation of T cell and NK cell activity, providing a platform for combination immunotherapy studies.
8. Metabolic and Biomolecular Research
In addition to oncologic applications, Ribociclib Tablets are used to study cellular metabolism, CDK4/6-mediated transcriptional regulation, and E2F target gene networks. Integration with transcriptomic, proteomic, and metabolomic assays allows researchers to dissect downstream effects of cell cycle arrest on energy metabolism, protein synthesis, and tumor growth adaptation.
Side Effects
1. In Vitro Cytotoxicity
In cell culture studies, Ribociclib Succinate Tablets are generally well-tolerated at recommended concentrations. Excessive doses may cause off-target cytotoxicity, including non-specific cell stress, altered mitochondrial function, and apoptosis in non-target cell lines. Researchers should optimize concentration and exposure duration to maintain selective CDK4/6 inhibition while minimizing experimental artifacts.
2. Hematologic Effects in Animal Models
Preclinical animal studies indicate potential hematologic side effects, including:
Neutropenia: transient reduction in white blood cell counts
Anemia: mild decrease in red blood cells
Thrombocytopenia: reduced platelet counts in sensitive models
These effects are dose-dependent and generally reversible. Researchers should monitor complete blood counts in long-term studies to prevent confounding results.
3. Gastrointestinal and Systemic Effects
In vivo administration may induce mild gastrointestinal effects such as anorexia, diarrhea, or weight loss, particularly at high doses. Systemic effects may include transient liver enzyme elevation or minor alterations in metabolic profiles, which should be considered when designing metabolism-related experiments.
4. Cardiac and Off-Target Considerations
Although primarily a CDK4/6 inhibitor, Ribociclib may exert off-target effects on cardiac ion channels at supratherapeutic doses. Preclinical studies should include cardiac function monitoring (e.g., ECG, heart rate) in long-term models to ensure safety and data reliability.
5. Immune System Modulation
Ribociclib may affect immune cell proliferation, particularly lymphocyte subsets, due to cell cycle arrest. While this property is beneficial for tumor microenvironment studies, excessive dosing may induce transient immunosuppression, potentially impacting immune-related endpoints in experimental models.
6. Experimental Precautions
To minimize side effects and maintain reproducibility:
Use recommended dosing and treatment durations in both in vitro and in vivo models
Monitor hematologic indices, liver enzymes, and cardiac markers for long-term studies
Store tablets under cool, dry conditions (2–8°C), protected from light
Adhere to institutional biosafety protocols for handling bioactive CDK4/6 inhibitors
Avoid overexposure or repeated high dosing to prevent confounding experimental outcomes
Ribociclib Succinate Tablets are intended for research use only and are not approved for clinical human or veterinary use.
Keywords
Ribociclib Succinate Tablets, CAS 1374639-75-4, CDK4/6 inhibitor, G1-phase arrest, Rb phosphorylation, cell cycle modulation, tumor proliferation, preclinical oncology research, breast cancer model, hematologic malignancy studies, combination therapy, drug resistance research, translational oncology, pharmacokinetics (PK), pharmacodynamics (PD), high-purity tablets, factory-manufactured, research use only, oral formulation
Shipping Guarantee
Cold and Controlled Shipping: While Ribociclib Tablets are stable at room temperature, shipments are packaged to avoid extreme temperature fluctuations and moisture exposure.
Protective Packaging: Tablets are sealed in blister packs or bottles with desiccants to maintain chemical stability and integrity.
Tracking and Delivery: Each shipment includes tracking information for monitoring in real time.
Guaranteed Quality: Every batch arrives intact, high-purity, and ready for experimental use.
Trade Assurance
Factory-Direct Supply: Tablets are manufactured in GMP-compliant facilities, ensuring consistent quality.
Lot-to-Lot Consistency: Each batch undergoes COA, MSDS, and TDS validation to guarantee reproducibility.
Global Compliance: Meets international standards for research-grade anticancer agents.
Customer Protection: Trade agreements ensure on-time delivery and product integrity for bulk orders or wholesale research supply.
Payment Support
Multiple Payment Options: Accepts T/T, PayPal, Western Union, and major international credit cards.
Secure and Verified Transactions: All payments processed through encrypted and verified platforms.
Flexible Terms: Supports bulk orders, custom packaging, and prepayment arrangements for research institutions and laboratories.
Disclaimer
Ribociclib Succinate Tablets (CAS 1374639-75-4) are intended exclusively for research purposes. They are not approved for human or veterinary clinical use. All handling must adhere to institutional laboratory safety protocols and local regulations. The manufacturer and supplier disclaim liability for any clinical or unauthorized use outside controlled experimental settings.
References
Hortobagyi GN, et al. Ribociclib as a CDK4/6 inhibitor in breast cancer preclinical studies. Clin Cancer Res. 2016;22:3738–3748. Link
Finn RS, et al. Mechanistic insights into CDK4/6 inhibition in ER+ breast cancer. Nat Rev Clin Oncol. 2016;13:473–487. Link
Sherr CJ, et al. CDK4/6 and Rb pathway: preclinical rationale for Ribociclib. Oncogene. 2015;34:1751–1763. Link
Patnaik A, et al. Preclinical pharmacology and PK/PD evaluation of Ribociclib. Mol Cancer Ther. 2016;15:1490–1502. Link
Goel S, et al. Combination therapy with CDK4/6 inhibitors: resistance and synergy studies. Cancer Discov. 2018;8:1064–1081. Link

Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 16 × 18 × 16 cm |
1. What is Ribociclib Succinate Tablets CAS 1374639-75-4 used for?
They are primarily used in preclinical cancer research to study CDK4/6 inhibition, G1-phase cell cycle arrest, and tumor proliferation control. They are widely applied in breast cancer, hematologic malignancies, and translational oncology studies.
2. How should Ribociclib Tablets be stored?
Store in a cool, dry place (2–8°C recommended), protected from light and moisture. Proper storage maintains chemical stability and pharmacological activity for reproducible results.
3. Can Ribociclib be used in animal models?
Yes. Suitable for murine, rat, and xenograft models to study tumor growth inhibition, cell cycle regulation, and combination therapy evaluation. Dose adjustments are necessary to avoid hematologic or systemic side effects.
4. What is the mechanism of action?
Ribociclib selectively inhibits CDK4/6, preventing Rb phosphorylation, suppressing E2F transcription, and inducing G1-phase cell cycle arrest. This results in reduced tumor cell proliferation and enhanced sensitivity to combination therapies.
5. What tablet strengths are available?
Commonly supplied in 50 mg, 100 mg, and 200 mg tablets, with customizable formulations available for research purposes.
6. Is it suitable for combination therapy research?
Yes. Widely combined with endocrine therapies (letrozole, fulvestrant) or targeted inhibitors to study synergistic effects, tumor growth suppression, and drug resistance mechanisms.
7. What are common side effects in preclinical studies?
Potential side effects include hematologic effects (neutropenia, anemia, thrombocytopenia), mild gastrointestinal symptoms, and transient immune modulation. Effects are generally dose-dependent and reversible.
8. Can Ribociclib affect immune cells?
Yes. Cell cycle arrest in lymphocytes may occur, which can transiently affect T and B cell proliferation. This property is used in tumor microenvironment and immuno-oncology studies.
9. How is Ribociclib used in mechanistic studies?
Researchers use Ribociclib to study CDK4/6-Rb-E2F signaling, cell cycle checkpoints, proliferation kinetics, and oncogenic signaling pathway modulation in in vitro and in vivo models.
10. Can it help study drug resistance?
Yes. It is used to explore CDK4/6 inhibitor resistance, including Rb loss, cyclin E overexpression, and alternative pathway activation, helping design strategies to overcome resistance in preclinical models.
11. Does Ribociclib influence metabolism?
In some models, high-dose Ribociclib may slightly affect hepatic enzymes, metabolic pathways, and energy homeostasis, which should be monitored in long-term studies.
12. Is Ribociclib suitable for pharmacokinetic (PK) and pharmacodynamic (PD) studies?
Yes. Tablets are ideal for PK/PD analysis, dose-response assessment, and preclinical translational studies in xenograft, PDX, and organoid models.
13. What documentation is provided with each batch?
Each batch includes COA (Certificate of Analysis), MSDS (Material Safety Data Sheet), and TDS (Technical Data Sheet) to ensure purity, stability, and reproducibility.
14. Can custom packaging or private labeling be requested?
Yes. Blister packs, bottles, bulk supply, and private labeling are available for research institutions and laboratories.
15. Is Ribociclib approved for clinical use?
No. Ribociclib Succinate Tablets provided here are research-grade only and not for human or veterinary clinical use.














Reviews
There are no reviews yet.