No products in the cart.
Ropivacaine Hydrochloride 100?mg / 10?mL Vial Wholesale price comparison
$1.00
Ropivacaine is a long-acting amide local anesthetic provided as a 1% (100?mg/10?mL) sterile solution. It offers effective sensory blockade with reduced motor impairment and lower cardiotoxicity risk, ideal for peripheral nerve block, epidural, and infiltration studies.
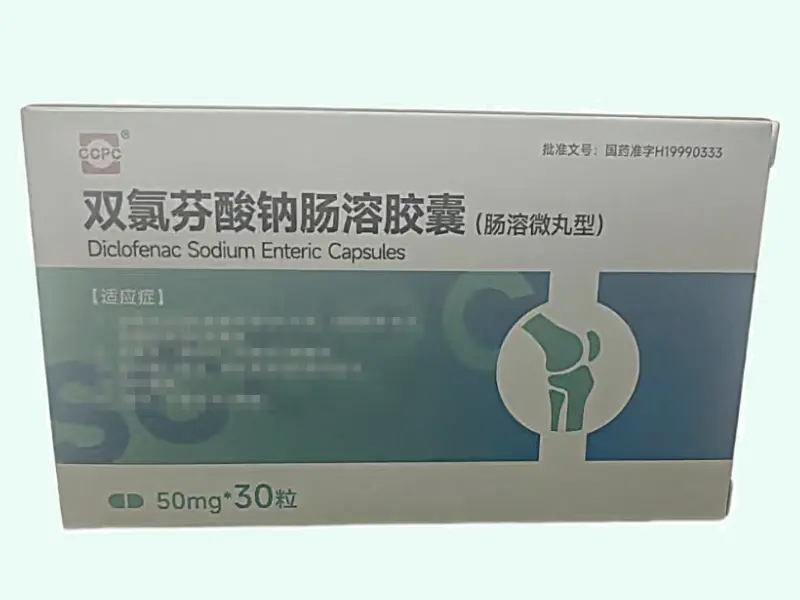
Manufactured by Chengdu Baiyu Pharmaceutical Co., Ltd. under approval H20233822, product code 86901012000784, barcode 6947954800208.
?? For laboratory research use only?Please consult staff for other specifications and uses?
Description
Product Specifications
| Attribute | Details |
|---|---|
| Product Name | Ropivacaine Hydrochloride Injection |
| Generic Name | Ropivacaine (S?enantiomer, HCl salt) |
| CAS Number | 84057?95?4 |
| Molecular Formula | C??H??N?O·HCl |
| Molecular Weight | ~274.4?g/mol |
| Dosage Form | Sterile aqueous solution for IV/IM/perineural injection |
| Strength | 100?mg per 10?mL vial (1%) |
| Pack Size | 1 vial per box |
| Approval Number | H20233822 (China NMPA) |
| Product Code | 86901012000784 |
| Manufacturer | Chengdu Baiyu Pharmaceutical Co., Ltd. |
| Barcode | 6947954800208 |
| Storage | Store at 2–25?°C; protect from light |
| Intended Use | Laboratory research only |
Mechanism of Action
Ropivacaine blocks voltage-gated sodium channels in neuronal membranes, inhibiting nerve impulse conduction. As a pure S-enantiomer, it provides effective sensory block with less motor block and reduced cardiotoxicity compared to bupivacaine meritpharm.com+15researchgate.net+15go.gale.com+15.
Research Applications & Pharmacology
Regional anesthesia models: Used in peripheral nerve block, epidural, spinal anesthesia, and surgical infiltration ncbi.nlm.nih.gov+3medicines.org.uk+3dailymed.nlm.nih.gov+3.
Pharmacokinetics:
Onset: ~10–45 minutes depending on route
Duration: up to 6–8 hours
Half-life: 1.6–6 hours
Metabolized in the liver (CYP1A2) and ?86% excreted renally en.wikipedia.org+3en.wikipedia.org+3zh.wikipedia.org+3
Safety Profile & Handling
Side effects: Minimal motor blockade; low risk of CNS symptoms (dizziness, tinnitus) or cardiovascular effects (hypotension, arrhythmia) .
Handling tips:
Administer under aseptic conditions.
Monitor for signs of systemic toxicity—stop if noted.
Have lipid emulsion therapy ready for overdose .
?? Research?Use Disclaimer
This product is intended solely for laboratory research. It is not approved for clinical, therapeutic, diagnostic, or veterinary applications. Misuse may pose health risks and compromise data integrity.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |










Reviews
There are no reviews yet.