No products in the cart.
Sale
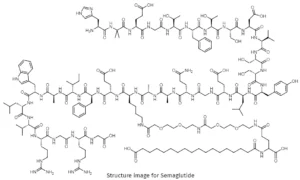
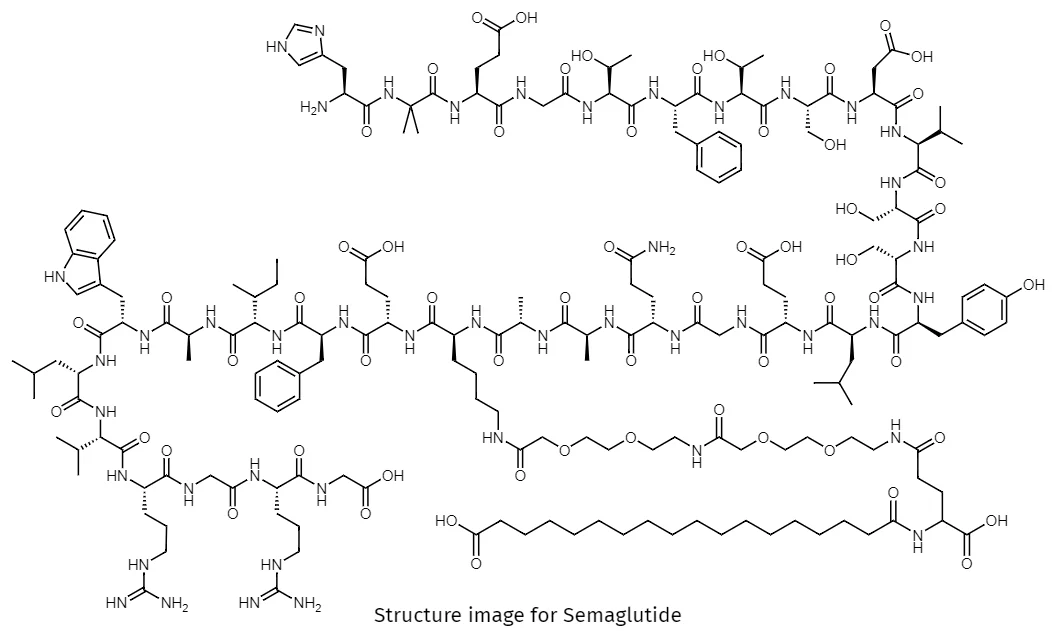
Semaglutide TFA – Purity 99.92% GMP Factory manufacture
Original price was: $36.00.$23.00Current price is: $23.00.
Semaglutide TFA is a high-purity, factory-manufactured research-grade peptide salt, supplied for in vitro molecular interaction studies, receptor signaling analysis, and biochemical mechanism investigations. Manufactured under strict quality control systems, this product supports academic laboratories, CROs, and industrial R&D teams requiring consistent quality, batch traceability, and scalable bulk supply.
Description
Product Description
Semaglutide TFA is a chemically well-defined peptide salt form engineered for advanced laboratory research applications, particularly in the fields of receptor–ligand interaction analysis, peptide structure–function relationships, and intracellular signaling pathway exploration. This product is strictly supplied for research use only, with all documentation and specifications tailored to professional experimental environments.
From a molecular research perspective, Semaglutide TFA serves as a model peptide ligand for studying GLP-1 receptor–associated signaling cascades, conformational stabilization strategies, and peptide modification effects at the molecular level. The trifluoroacetate (TFA) counterion contributes to controlled physicochemical characteristics, enabling reproducibility in in vitro assay systems and analytical workflows.
This material is produced via factory-scale synthesis, followed by multi-step purification and analytical verification, ensuring high purity, low batch-to-batch variability, and reliable experimental consistency. Each production batch undergoes identity confirmation and purity assessment prior to release, supporting reproducible outcomes across independent laboratories.
As a China-based B2B export product, Semaglutide TFA is widely supplied to research institutes, biotechnology companies, and pharmaceutical R&D organizations worldwide. Flexible bulk, wholesale, and customization options are available to support both exploratory research and long-term experimental programs.
Importantly, all descriptive content, specifications, and positioning of this product are strictly limited to laboratory research contexts, with no implication of administration, diagnostic application, or biological exposure beyond controlled experimental systems.

| Category | Specification Details |
|---|---|
| Product Name | Semaglutide TFA |
| Chemical Classification | Research-grade modified peptide (TFA salt form) |
| Molecular Nature | Long-chain stabilized peptide analog |
| Counterion | Trifluoroacetate (TFA) |
| Purity | ≥ 98% (HPLC, area normalization) |
| Identity Confirmation | Mass spectrometry (MS) and chromatographic profiling |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Hygroscopicity | Slightly hygroscopic under ambient laboratory conditions |
| Solubility Profile | Freely soluble in common laboratory aqueous buffers and compatible organic solvent systems used in analytical research |
| Molecular Weight | Provided per batch based on analytical verification |
| Structural Integrity | Verified peptide sequence consistency and modification pattern |
| Manufacturing Process | Factory-scale synthesis with multi-step purification |
| Quality Control Standards | Each batch tested for purity, identity, and consistency |
| Batch Traceability | Complete batch records available for research documentation |
| Stability | Stable under recommended controlled laboratory storage conditions |
| Storage Recommendation | Store in sealed containers under dry, temperature-controlled laboratory environments |
| Packaging Options | Vials or bottles suitable for laboratory handling |
| Available Quantities | Milligram to gram-scale |
| Supply Type | Bulk · Wholesale · Long-term research supply |
| Customization | OEM labeling, packaging, and specification alignment available |
| Country of Origin | China |
| Export Model | Professional B2B international research material supply |
| Intended Use | In vitro research, molecular mechanism studies, receptor interaction analysis |
| Regulatory Status | Supplied strictly as research-grade material |
Supplementary Notes on Specifications
Purity & Consistency
High-performance liquid chromatography (HPLC) is employed to ensure batch-to-batch reproducibility, supporting comparative and longitudinal experimental studies.Structural Reliability
Peptide sequence integrity and modification patterns are confirmed to maintain reliable molecular behavior in receptor and signaling pathway research.Scalability for Research Programs
Factory-manufactured production allows stable bulk supply, making Semaglutide TFA suitable for high-throughput screening, repeated assay cycles, and long-term R&D projects.B2B Research Compliance
All specifications are aligned with global laboratory research procurement standards, supporting export to universities, CROs, and industrial research organizations.
Mechanism of Action
Receptor–Ligand Recognition and Binding Dynamics
At the molecular level, Semaglutide TFA functions as a high-affinity peptide ligand model for studying glucagon-like peptide-1 receptor (GLP-1R)–associated binding mechanisms within controlled experimental systems. In vitro receptor-binding assays demonstrate that its modified peptide backbone promotes stable ligand–receptor complex formation, making it a valuable tool for analyzing recognition specificity, binding orientation, and interaction strength.
The presence of structural modifications within the peptide sequence allows researchers to investigate how ligand stabilization strategies influence receptor engagement, providing insights into molecular recognition rules governing class B G-protein–coupled receptors (GPCRs). These interactions are typically examined using receptor-expressing cell models or isolated receptor preparations under non-clinical laboratory conditions.
Conformational Stabilization and Structural Modulation
Semaglutide TFA exhibits enhanced conformational stability compared with unmodified peptide analogs, enabling detailed exploration of structure–function relationships. This stability supports reproducible analysis of secondary and tertiary structural features that influence receptor interaction and downstream signal initiation.
From a mechanistic research standpoint, Semaglutide TFA is frequently employed to evaluate how peptide backbone rigidity, side-chain orientation, and sequence modification affect molecular alignment within receptor binding domains. These studies contribute to broader understanding of peptide engineering principles and receptor–ligand co-evolution.
Intracellular Signaling Cascade Initiation (In Vitro Models)
Upon receptor engagement in experimental systems, Semaglutide TFA serves as a reference ligand for mapping GLP-1R–associated intracellular signaling pathways. In vitro signaling assays allow researchers to characterize second-messenger modulation, pathway selectivity, and signal amplitude without extending beyond laboratory research boundaries.
Such mechanistic investigations help delineate signal transduction efficiency and pathway bias, supporting comparative studies between peptide analogs and aiding in the interpretation of receptor activation profiles at the molecular level.
Receptor Desensitization and Recycling Studies
Semaglutide TFA is also applied in mechanistic studies of receptor regulation, including receptor internalization, recycling, and signaling attenuation under controlled experimental conditions. These investigations focus on understanding how ligand structural features influence receptor trafficking behavior and signal persistence in vitro.
By serving as a standardized ligand reference, Semaglutide TFA enables reproducible examination of temporal signaling dynamics, contributing to mechanistic datasets relevant to receptor biology and molecular pharmacology research.
Role in Structure–Activity Relationship (SAR) Research
As a well-characterized peptide ligand, Semaglutide TFA is widely used in structure–activity relationship (SAR) studies to correlate chemical modification patterns with observed molecular signaling responses. Researchers utilize these data to refine predictive models of peptide–receptor interaction and to inform rational design strategies in experimental peptide chemistry.
These SAR-focused investigations are strictly confined to in vitro analytical frameworks, emphasizing molecular insight rather than application-oriented outcomes.
Integration with Computational and Systems-Level Analysis
Mechanistic data generated using Semaglutide TFA are frequently integrated with computational modeling, molecular docking, and simulation studies. This integration supports in silico validation of experimental observations, allowing researchers to visualize ligand–receptor interfaces and predict interaction energetics.
When combined with multi-omic and systems biology approaches, Semaglutide TFA serves as a reference molecular probe for advancing mechanistic understanding of GPCR-mediated signaling networks in a purely research-oriented context.
Summary of Mechanistic Value
In summary, Semaglutide TFA provides a robust and reproducible molecular framework for investigating:
Ligand–receptor binding mechanisms
Peptide conformational stability and structural modulation
In vitro signal transduction initiation and regulation
Receptor trafficking and signaling dynamics
Structure–activity relationships and computational modeling
All mechanistic interpretations remain strictly within laboratory research and experimental analysis domains, reinforcing its role as a high-purity, factory-manufactured research-grade peptide for advanced molecular studies.
Applications
Receptor–Ligand Interaction Research
Semaglutide TFA is widely applied in receptor–ligand interaction studies focusing on glucagon-like peptide-1 receptor (GLP-1R) systems under controlled laboratory conditions. As a structurally stabilized peptide ligand, it supports detailed evaluation of binding affinity, interaction specificity, and ligand orientation using receptor-expressing experimental platforms or isolated receptor preparations. These applications enable precise characterization of molecular recognition mechanisms central to GPCR research.
In Vitro Signal Transduction Analysis
In vitro signaling assays represent a core application area for Semaglutide TFA. Researchers employ this material to investigate receptor-associated intracellular signaling cascades, including pathway activation profiles, signal amplification behavior, and temporal response characteristics. Such studies provide insight into signal selectivity and pathway bias, contributing to mechanistic mapping of receptor-mediated communication at the molecular level.
Structure–Activity Relationship (SAR) Studies
Semaglutide TFA serves as a reference compound in structure–activity relationship (SAR) research, allowing systematic comparison between modified peptide analogs. By correlating structural features with observed molecular responses, researchers gain deeper understanding of peptide backbone modification, side-chain contributions, and conformational stability. These applications are central to experimental peptide chemistry and rational molecular design research.
Analytical Method Development and Validation
The high purity and batch consistency of Semaglutide TFA make it suitable for analytical method development, including chromatographic profiling, stability assessment, and comparative analytical validation studies. Laboratories use it to establish robust analytical workflows and to benchmark assay sensitivity and reproducibility under standardized research conditions.
Computational Modeling and Systems Biology Integration
Semaglutide TFA is frequently incorporated into computational modeling and in silico simulation studies, such as molecular docking, dynamic simulations, and receptor–ligand interface visualization. When combined with experimental datasets, it supports systems-level analysis of receptor signaling networks, enhancing mechanistic interpretation within multi-omic and data-driven research frameworks.
Long-Term and High-Throughput Research Programs
Owing to its factory-manufactured consistency and bulk availability, Semaglutide TFA is well-suited for long-term experimental programs and high-throughput screening workflows. These applications are particularly relevant for CROs, academic laboratories, and industrial R&D teams requiring stable supply, reproducible performance, and scalable research materials.
Research Models
Semaglutide TFA is compatible with a wide range of non-clinical research models, including:
Receptor-expressing cell lines
Cell-free biochemical assay platforms
Molecular docking and computational modeling systems
High-throughput screening assay formats
All models are strictly experimental and non-diagnostic in nature.
Experimental Design Considerations
When incorporating Semaglutide TFA into experimental workflows, researchers should consider purity verification, analytical method alignment, and controlled environmental parameters. Consistency in assay conditions supports data comparability across batches and research sites.
It is recommended to document batch numbers, analytical methods, and experimental configurations to ensure traceability and reproducibility in long-term research projects.

Laboratory Safety & Handling Guidelines
Semaglutide TFA is supplied strictly as a research-grade chemical material and should be handled exclusively within professional laboratory environments by trained personnel familiar with standard chemical and biochemical safety protocols. All handling activities must comply with institutional laboratory regulations, internal SOPs, and applicable chemical safety standards.
General Laboratory Handling Practices
Personnel handling Semaglutide TFA should wear appropriate personal protective equipment (PPE), including laboratory gloves, protective clothing, and eye protection where required. Direct contact with the material should be avoided, and all procedures should be performed on clean, designated laboratory benches or within appropriate containment systems to minimize environmental contamination.
Handling should be conducted using clean, dry laboratory tools, ensuring that cross-contamination with other research materials is prevented. Dedicated instruments and containers are recommended for peptide-related experimental workflows to maintain material integrity and analytical accuracy.
Storage Conditions and Material Stability
Semaglutide TFA should be stored in sealed, clearly labeled containers under controlled laboratory conditions, away from moisture, direct light, and sources of contamination. Maintaining stable environmental parameters supports long-term material stability and reproducibility in experimental results.
Repeated exposure of containers to ambient laboratory air should be minimized. Where applicable, aliquoting under controlled conditions may be employed to reduce unnecessary handling cycles and preserve sample consistency across experiments.
Preparation and Transfer Procedures
All preparation, weighing, and transfer steps should be carried out using analytical-grade laboratory equipment. Workflows should be designed to minimize material loss and prevent aerosolization or dispersion of fine particles. Surfaces and instruments should be cleaned following completion of procedures in accordance with laboratory cleaning protocols.
Clear documentation of preparation steps, including batch identification and handling history, is recommended to support traceability and experimental reproducibility.
Waste Management and Disposal
Any waste generated during handling or experimentation involving Semaglutide TFA should be managed as research laboratory chemical waste. Disposal must follow institutional guidelines and local regulatory requirements for non-clinical chemical materials. Mixing with incompatible waste streams should be avoided.
Containers, consumables, and residual material should be rendered non-reusable where required by laboratory policy and disposed of through approved waste management channels.
Risk Management and Documentation
Although supplied as a high-purity research material, Semaglutide TFA should be treated with the same level of caution applied to specialty laboratory chemicals. Laboratories are encouraged to maintain internal risk assessments, handling records, and safety documentation associated with its use.
Proper training, documentation, and adherence to established safety frameworks ensure safe handling, regulatory compliance, and integrity of experimental research outcomes.
Summary
By implementing standardized laboratory safety and handling practices, Semaglutide TFA can be integrated smoothly into advanced in vitro research workflows. Adherence to these guidelines supports personnel safety, material stability, and data reliability, reinforcing its role as a factory-manufactured, high-purity research-grade peptide for professional laboratory use.
Integration with Multi-Omic & Computational Studies
Semaglutide TFA is well suited for integration into multi-omic research frameworks and computational analysis pipelines designed to investigate receptor-mediated molecular systems under controlled experimental conditions. As a structurally well-characterized peptide ligand, it provides a reliable reference point for correlating transcriptomic, proteomic, and metabolomic datasets generated in receptor-expressing in vitro models.
Within transcriptomic and proteomic studies, Semaglutide TFA supports the systematic analysis of signal-associated gene expression patterns and protein interaction networks, enabling researchers to map pathway-level responses and molecular cross-talk. These datasets contribute to a deeper understanding of receptor-regulated signaling architectures at the systems level.
From a computational perspective, Semaglutide TFA is frequently incorporated into molecular docking, dynamic simulation, and receptor–ligand interface modeling workflows. In silico approaches allow visualization of binding conformations, interaction energetics, and structural stability, complementing experimental observations and enhancing mechanistic interpretation.
When combined with data-driven modeling, network analysis, and machine learning–assisted prediction tools, Semaglutide TFA facilitates integrative studies aimed at identifying pattern consistency, pathway bias, and molecular interaction trends. This integration strengthens reproducibility, supports hypothesis generation, and advances systems-level understanding of peptide–receptor interactions within non-clinical research environments.

Keywords
Semaglutide TFA, Semaglutide TFA high purity, Semaglutide TFA research grade, Semaglutide TFA wholesale, Semaglutide TFA bulk supply, Semaglutide TFA factory manufactured, peptide research material, GLP-1 receptor ligand research, in vitro peptide mechanism study, China B2B peptide export
Shipping Guarantee
Secure, research-grade packaging is used to protect material integrity throughout international transit and handling processes. Each shipment is prepared in accordance with laboratory material export standards, ensuring resistance to environmental fluctuations encountered during transportation. Export logistics are optimized for global laboratory delivery, supporting predictable transit timelines and consistent arrival conditions. Accompanying documentation is systematically prepared to facilitate customs clearance, research material verification, and internal laboratory receipt procedures. This integrated shipping approach supports uninterrupted research workflows for international partners.

Trade Assurance
All products are supplied through factory-direct manufacturing channels, ensuring full control over production quality and documentation accuracy. Verified batch traceability and analytical records are maintained to support research reproducibility and internal audit requirements. Long-term bulk and wholesale cooperation is available for CROs, academic research institutes, and industrial R&D organizations. Stable production capacity enables consistent supply for extended research programs and multi-phase experimental projects. Trade assurance frameworks are designed to support transparent, reliable, and scalable B2B collaboration.

Payment Support
Multiple payment methods are supported to accommodate international B2B procurement requirements. Credit Card payments provide convenience and speed for standard research orders. T/T (Telegraphic Transfer) options are available for bulk, wholesale, and long-term cooperation arrangements. Cryptocurrency payments are supported through encrypted transaction channels, offering an additional secure and flexible settlement option. All payment processes are handled under standardized commercial and compliance procedures.
Disclaimer
This product is supplied strictly for laboratory research use only. It is not intended for diagnostic, therapeutic, or any non-research-related applications. All technical descriptions, specifications, and references are provided solely for scientific, educational, and experimental purposes. Any use must be conducted within controlled laboratory environments by qualified professionals. The supplier assumes no responsibility for applications beyond the defined research scope.
References
National Center for Biotechnology Information (NCBI) – Semaglutide overview
https://pubchem.ncbi.nlm.nih.govRCSB Protein Data Bank – GLP-1 receptor structural data
https://www.rcsb.orgIUPAC – Peptide nomenclature and standards
https://iupac.orgFDA Drug Substance Information (for chemical reference only)
https://www.fda.govEuropean Bioinformatics Institute (EMBL-EBI) – Molecular interaction databases
https://www.ebi.ac.uk
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
Q1: What is Semaglutide TFA intended for?
A1: Semaglutide TFA is supplied strictly as a research-grade peptide material intended for laboratory-based studies, including in vitro receptor interaction analysis, molecular mechanism exploration, and biochemical research applications.
Q2: Is this product suitable for in vitro experimental systems?
A2: Yes. Semaglutide TFA is specifically manufactured for controlled in vitro research environments, supporting reproducible experimental workflows and analytical studies.
Q3: Does Semaglutide TFA meet research-grade purity standards?
A3: Each batch is produced under factory-controlled conditions and verified to meet high-purity research-grade specifications, typically ≥98% by HPLC analysis.
Q4: How is batch consistency ensured?
A4: Batch-to-batch consistency is maintained through standardized synthesis, purification, and quality control procedures, with batch traceability and analytical documentation available for research records.
Q5: Can this product be used as a reference compound in mechanistic studies?
A5: Yes. Due to its well-characterized structure and stability, Semaglutide TFA is commonly used as a reference ligand in molecular mechanism and signaling pathway research.
Q6: Is Semaglutide TFA compatible with receptor-binding assays?
A6: It is frequently applied in receptor–ligand binding and interaction studies under non-clinical laboratory conditions.
Q7: Can this product support long-term research projects?
A7: Yes. Factory-scale manufacturing and stable supply capacity make it suitable for long-term, multi-phase research programs requiring consistent materials.
Q8: Are bulk and wholesale quantities available?
A8: Bulk and wholesale supply options are available to support CROs, academic laboratories, and industrial R&D organizations.
Q9: Is customization or OEM service supported?
A9: Customization options, including packaging, labeling, and specification alignment, can be discussed for qualified B2B research procurement needs.
Q10: What documentation can be provided with the product?
A10: Documentation may include batch identification, purity analysis summaries, and quality-related records, depending on project requirements.
Q11: How should Semaglutide TFA be stored in a laboratory setting?
A11: It should be stored in sealed, clearly labeled containers under controlled laboratory conditions, following institutional storage guidelines for research chemicals.
Q12: Is this product suitable for computational or in silico studies?
A12: Yes. Structural and analytical data associated with Semaglutide TFA make it suitable for molecular docking, simulation, and computational modeling research.





















Reviews
There are no reviews yet.