No products in the cart.
Sale
Trifluridine/Tipiracil Tablets 150mg | CAS 70-00-8 | Thymidine Analog for Antiviral and Anticancer Research
Original price was: $3.00.$2.00Current price is: $2.00.
Trifluridine (CAS 70-00-8) is a fluorinated pyrimidine nucleoside analog structurally related to thymidine. It is widely used in antiviral and anticancer research to study DNA synthesis inhibition, viral replication interference, and fluoropyrimidine-based chemotherapeutic mechanisms in preclinical models.
Description
Contents
hide
Product Description
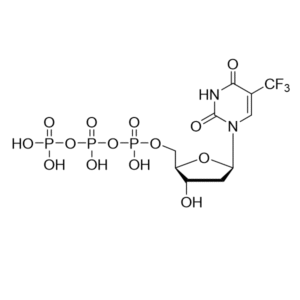
Trifluridine, also known as 5-trifluoromethyl-2′-deoxyuridine, is a thymidine analog in which the methyl group on the pyrimidine ring is substituted by a trifluoromethyl group. This modification gives Trifluridine potent biochemical activity as an inhibitor of DNA synthesis.
In research, Trifluridine is primarily used to:
Study the mechanisms of DNA polymerase inhibition.
Explore nucleoside analog incorporation into viral or cancer cell DNA.
Investigate fluoropyrimidine-induced cytotoxicity and replication stress in proliferating cells.
Model antiviral and anticancer nucleoside metabolism.
Mechanistically, Trifluridine undergoes phosphorylation to its active triphosphate form (TF-TTP) by thymidine kinase (TK). The triphosphate derivative competes with thymidine triphosphate for incorporation into DNA, leading to base mispairing, DNA strand breaks, and inhibition of DNA synthesis. In viral research, Trifluridine demonstrates inhibitory activity against DNA viruses, such as herpes simplex virus (HSV), by interfering with viral DNA polymerase function.
In oncology research, Trifluridine is a key component of the combination drug trifluridine/tipiracil, designed to improve stability and bioavailability in preclinical and clinical models. This pairing enables sustained exposure to the active metabolite, facilitating studies of nucleoside analog synergy, DNA repair response, and tumor proliferation control.
Product Specifications
| Item | Specification |
|---|---|
| Product Name | Trifluridine |
| CAS Number | 70-00-8 |
| Synonyms | 5-Trifluoromethyl-2′-deoxyuridine; F3TDR; Trifluorothymidine |
| Molecular Formula | C10H11F3N2O5 |
| Molecular Weight | 296.20 g/mol |
| Purity | ≥99% |
| Appearance | White to off-white crystalline powder |
| Solubility | Soluble in DMSO, ethanol, and water |
| Storage Temperature | 2–8 °C |
| Category | Nucleoside analog, antiviral and anticancer research reagent |
| Applications | Antiviral mechanism studies, DNA synthesis inhibition assays, fluoropyrimidine cytotoxicity research |
| Formulation | Lyophilized or crystalline form for in vitro research |
| Stability | Stable under recommended storage conditions |
| Shelf Life | 24 months |
| Supplier Type | Research chemical manufacturer |
| Intended Use | For laboratory research use only |
Mechanism of Action
Trifluridine’s biological activity is derived from its mimicry of thymidine, allowing incorporation into DNA during replication.
1. Phosphorylation and Activation
Trifluridine is converted intracellularly by thymidine kinase to its monophosphate, diphosphate, and triphosphate derivatives. The active form, TF-TTP, competes with thymidine triphosphate for DNA polymerase-mediated incorporation.
2. DNA Incorporation and Misincorporation
Once incorporated, the trifluoromethyl group alters base pairing and hydrogen bonding, disrupting DNA replication fidelity. This process triggers replication fork collapse and DNA damage signaling pathways.
3. Inhibition of DNA Synthesis
Through competitive inhibition, Trifluridine reduces DNA chain elongation and overall DNA synthesis rates, particularly in rapidly dividing or virally infected cells.
Trifluridine inhibits viral DNA polymerase more effectively than host enzymes, leading to suppression of viral replication — especially in herpes simplex virus (HSV) studies.
5. Research Applications
Mechanistic studies of fluorinated nucleosides.
Analysis of DNA polymerase kinetics and fidelity.
Examination of DNA repair pathways and replication stress markers.
Evaluation of antiviral and cytotoxic synergy with other nucleoside analogs or polymerase inhibitors.

Side Effects
In preclinical or in vitro studies, Trifluridine exposure can lead to:
DNA strand breaks and replication arrest.
Cytotoxicity in rapidly dividing cells.
Altered mitochondrial DNA synthesis at high concentrations.
Oxidative stress and p53 pathway activation.
These effects are valuable for mechanistic exploration but emphasize that Trifluridine is for laboratory research use only, not for therapeutic or diagnostic purposes.
Keywords
Trifluridine, CAS 70-00-8, fluorinated thymidine analog, nucleoside analog research, antiviral compound, anticancer nucleoside, DNA synthesis inhibitor, fluoropyrimidine study, viral polymerase inhibitor, DNA incorporation research, high-purity nucleoside analog, OEM & bulk nucleoside supplier China.
Shipping Guarantee
All shipments are handled using validated cold-chain logistics to preserve compound integrity. Each package is sealed in moisture-proof containers with secondary protective wrapping and continuous temperature monitoring. Products are shipped via express international couriers with full tracking and insurance coverage.
Trade Assurance
We ensure product authenticity, verified ≥99% purity, and compliance with analytical standards (HPLC, MS, and NMR). Each batch is supplied with a Certificate of Analysis (CoA). Our trade assurance policy guarantees replacement or refund for any deviation from listed specifications.
Payment Support
We provide flexible and secure global payment options to support international research transactions. Accepted payment methods include PayPal, major credit cards (Visa, MasterCard, American Express), telegraphic transfer (T/T), and cryptocurrencies (USDT, Bitcoin, Ethereum). All transactions are protected by industry-standard encryption and verified payment gateways to ensure confidentiality and fund security.
Disclaimer
All products listed are intended for laboratory research use only and not for human or veterinary use. They are not drugs, medical devices, or diagnostics and should not be administered to humans or animals. Researchers must handle all materials in accordance with institutional biosafety and chemical safety guidelines. The information provided is for scientific reference only and does not imply therapeutic efficacy, safety, or regulatory approval.
Additional information
| Weight | 1.1 kg |
|---|---|
| Dimensions | 18 × 16 × 18 cm |
What is Trifluridine?
Trifluridine is a fluorinated thymidine analog used in research to study DNA synthesis inhibition and nucleoside incorporation mechanisms.
What is the CAS number of Trifluridine?
The CAS number is 70-00-8.
What is the purity level of the research-grade compound?
Each batch is verified at ≥99% purity through HPLC and NMR analysis.
Is Trifluridine used in antiviral research?
Yes, it is widely used to study viral replication inhibition, especially against herpes simplex virus (HSV).
Can Trifluridine be applied to cancer research?
Yes, it serves as a fluoropyrimidine analog model for studying DNA synthesis inhibition and tumor cell cytotoxicity.
How is it activated intracellularly?
Trifluridine is phosphorylated by thymidine kinase to its active triphosphate form (TF-TTP).
What solvent is recommended for dissolution?
It is soluble in DMSO, ethanol, and water under research conditions.
What is the recommended storage temperature?
Store at 2–8 °C in tightly sealed containers.
Does Trifluridine have specificity for viral polymerases?
Yes, it exhibits greater affinity for viral DNA polymerases than host enzymes.
Can it be combined with other compounds?
It is often co-studied with tipiracil and other nucleoside analogs for synergistic effects.
Is OEM and bulk supply available?
Yes, OEM and bulk research-grade production are available through our peptide and small-molecule manufacturing facilities in China.
Does it come with a Certificate of Analysis?
Yes, each order includes a CoA verifying purity, identity, and analytical data.
What category does this compound belong to?
It is classified as a nucleoside analog and antiviral/anticancer research reagent.
Can I order Trifluridine online?
Yes, it is available for secure international ordering through our online research chemical platform.
Is Trifluridine suitable for in vivo use?
No, it is strictly intended for laboratory research and in vitro applications only.
















Reviews
There are no reviews yet.