No products in the cart.
Aureobasidin A – 99%High-purity peptides
$2.00
Aureobasidin A is a highly specific cyclic depsipeptide inhibitor of inositol phosphorylceramide (IPC) synthase, widely applied in in vitro studies of sphingolipid biosynthesis, membrane lipid regulation, and fungal cell membrane signaling mechanisms.
Description
Contents
hide
Product Description
Aureobasidin A is a highly specific cyclic depsipeptide inhibitor that plays a critical role in in vitro investigations of sphingolipid biosynthesis and membrane lipid regulation. Originally isolated from the fungus Aureobasidium pullulans, Aureobasidin A has become a widely recognized molecular research tool due to its exceptional selectivity toward inositol phosphorylceramide (IPC) synthase, a key enzymatic component within sphingolipid metabolic pathways.
Sphingolipids are essential structural and functional elements of biological membranes, influencing membrane organization, lipid microdomain formation, and lipid-dependent signaling cascades. IPC synthase catalyzes the conversion of ceramide into inositol phosphorylceramide, a pivotal step that regulates sphingolipid flux and maintains lipid homeostasis. Aureobasidin A enables researchers to selectively disrupt this single enzymatic node, allowing precise dissection of pathway-specific biochemical mechanisms under controlled experimental conditions.
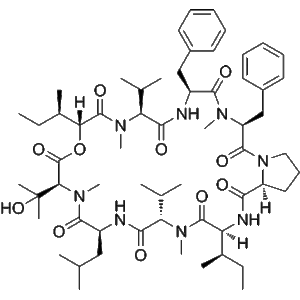
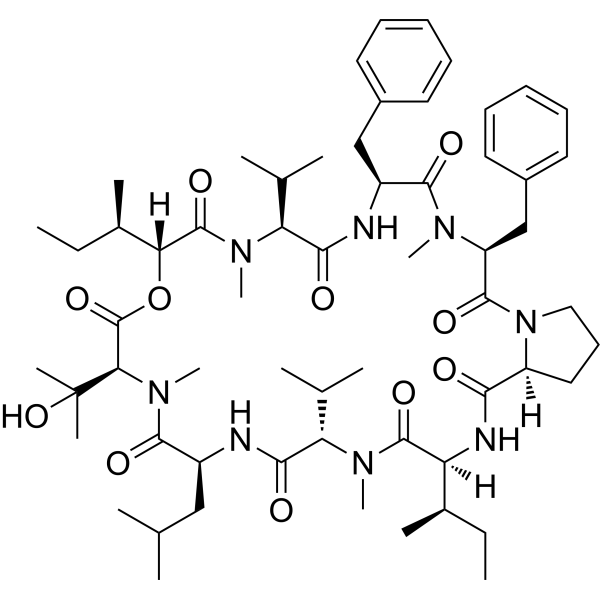
From a structural perspective, Aureobasidin A features a macrocyclic depsipeptide architecture that confers both stability and high-affinity interaction with its molecular target. This unique configuration supports reproducible binding behavior, making the compound particularly suitable for enzyme inhibition assays, lipid pathway mapping, and structure–activity relationship (SAR) analysis. Its well-characterized molecular profile has also facilitated extensive use in computational docking studies and molecular dynamics simulations, providing mechanistic insight at the atomic level.
In laboratory research settings, Aureobasidin A is frequently applied to explore ceramide accumulation dynamics, sphingolipid turnover rates, and lipid-mediated regulatory processes. By selectively inhibiting IPC synthesis, researchers can monitor downstream alterations in membrane lipid composition and assess the functional consequences of sphingolipid imbalance. These investigations are essential for understanding how lipid metabolism influences membrane integrity, protein localization, and intracellular signaling networks in simplified experimental systems.
This product is manufactured under strict quality control standards, ensuring high chemical purity and batch-to-batch consistency. The availability of factory-direct Aureobasidin A supports scalable research demands, from exploratory laboratory studies to large-volume screening programs. Bulk supply, low-cost wholesale options, and China-based B2B export capabilities make this compound accessible to academic institutions, biotechnology companies, and pharmaceutical research organizations worldwide.
Importantly, Aureobasidin A is supplied exclusively for laboratory research use, with applications limited to in vitro biochemical, biophysical, lipidomic, and computational studies. Its defined mechanism, high specificity, and reproducible performance make it a reliable molecular probe for advanced investigations into sphingolipid biosynthesis and membrane lipid regulation, supporting both fundamental research and pathway-focused discovery programs.
Product Specifications
The following specifications define the physicochemical and quality attributes of Aureobasidin A supplied for professional laboratory research applications. All parameters are established to support in vitro biochemical, lipidomic, and molecular mechanism studies, with an emphasis on consistency, traceability, and experimental reproducibility.
| Parameter | Specification |
|---|---|
| Product Name | Aureobasidin A |
| Chemical Class | Cyclic depsipeptide |
| CAS Number | 127785-64-2 |
| Molecular Formula | C₆₀H₉₂N₈O₁₆ |
| Molecular Weight | ~1101.3 g/mol |
| Purity | ≥98–99% (HPLC-verified) |
| Appearance | White to off-white solid powder |
| Solubility Profile | Soluble in DMSO; limited solubility in aqueous buffers |
| Stability | Stable under recommended storage conditions |
| Storage Conditions | −20 °C, dry environment, light-protected |
| Supply Form | Solid powder |
| Quality Documentation | Certificate of Analysis (COA), MSDS |
Each production batch of Aureobasidin A undergoes high-performance liquid chromatography (HPLC) analysis to confirm purity and identity, ensuring suitability for enzyme inhibition assays and sphingolipid pathway studies. Molecular weight and structural integrity are further verified using spectrometric techniques, supporting reliable downstream interpretation of experimental data.
The solubility characteristics of Aureobasidin A allow controlled preparation of stock solutions for precise concentration-dependent studies, minimizing variability in experimental outcomes. Proper storage at low temperature and protection from light are recommended to maintain chemical stability during long-term research use.
This product is factory manufactured in China under standardized quality management systems, enabling bulk production, low-cost wholesale supply, and global B2B export. Comprehensive documentation, including COA and MSDS, is provided with each shipment to support regulatory compliance and internal laboratory quality assurance processes.
Aureobasidin A supplied under these specifications is intended exclusively for laboratory research use, ensuring reliable performance in advanced sphingolipid biosynthesis, membrane lipid regulation, and molecular mechanism investigations.
Mechanism of Action
Aureobasidin A functions as a highly selective inhibitor of inositol phosphorylceramide (IPC) synthase, an essential enzyme within the sphingolipid biosynthesis pathway. IPC synthase catalyzes the transfer of a phosphoinositol moiety from phosphatidylinositol to ceramide, forming inositol phosphorylceramide, a critical sphingolipid intermediate involved in membrane lipid organization and pathway regulation. By targeting this enzymatic step with high specificity, Aureobasidin A enables precise modulation of sphingolipid metabolic flux in controlled in vitro research systems.
At the molecular level, Aureobasidin A interacts directly with IPC synthase through its macrocyclic depsipeptide structure, which provides a conformationally constrained binding interface. This architecture facilitates stable association with the enzyme’s active or regulatory regions, effectively blocking catalytic turnover without broadly interfering with unrelated lipid biosynthesis enzymes. As a result, Aureobasidin A demonstrates a high degree of pathway selectivity, making it an ideal molecular probe for mechanistic studies that require minimal off-target effects.
Inhibition of IPC synthase by Aureobasidin A leads to a reduction in IPC formation and a corresponding accumulation of upstream ceramide intermediates. This controlled alteration of sphingolipid composition allows researchers to investigate how ceramide levels influence membrane structure, lipid–protein interactions, and lipid-dependent signaling processes. Because sphingolipids contribute to membrane microdomain formation, Aureobasidin A-mediated pathway interruption provides insight into the dynamic relationship between lipid biosynthesis and membrane organization.
Beyond direct enzyme inhibition, Aureobasidin A indirectly affects downstream lipid regulatory networks. Changes in sphingolipid balance can influence the distribution and function of membrane-associated proteins, alter lipid packing properties, and modulate biochemical signaling cascades that are sensitive to membrane composition. These secondary effects are particularly valuable for dissecting structure–function relationships within lipid-regulated systems, when studied under defined experimental conditions.
Aureobasidin A has also been extensively utilized in computational and structural biology studies. Its well-defined chemical structure supports enzyme–ligand docking simulations, molecular dynamics modeling, and structure–activity relationship analyses. These approaches complement biochemical data by providing atomistic insight into binding interactions and inhibitory mechanisms, further enhancing understanding of IPC synthase regulation.
Through its selective inhibition of IPC synthase and predictable biochemical consequences, Aureobasidin A serves as a precise molecular tool for advanced investigations into sphingolipid biosynthesis, membrane lipid regulation, and lipid-dependent molecular mechanisms in in vitro laboratory research environments.
Applications
Aureobasidin A is widely applied as a specialized molecular research tool in in vitro studies focused on sphingolipid biosynthesis, membrane lipid regulation, and enzyme-specific pathway analysis. Its high selectivity for inositol phosphorylceramide (IPC) synthase makes it particularly valuable for experimental designs that require precise and controllable modulation of sphingolipid metabolic flux without broadly perturbing unrelated lipid pathways.
One primary application of Aureobasidin A is in biochemical enzyme inhibition assays, where it is used to characterize IPC synthase activity, assess catalytic dynamics, and evaluate pathway responsiveness under defined experimental conditions. These studies support quantitative analysis of enzyme kinetics, inhibition profiles, and concentration-dependent effects, providing foundational data for lipid metabolism research.
Aureobasidin A is also extensively utilized in sphingolipid pathway mapping and lipidomics-based investigations. By selectively blocking IPC formation, researchers can monitor changes in ceramide levels and related lipid intermediates, enabling detailed profiling of sphingolipid distribution and turnover. Integration with mass spectrometry–based lipidomics allows high-resolution assessment of pathway-specific lipid alterations.
In membrane lipid organization studies, Aureobasidin A serves as a tool to explore the relationship between sphingolipid biosynthesis and membrane structural properties. Controlled modulation of IPC synthesis facilitates examination of lipid microdomain formation, membrane packing behavior, and lipid–protein interaction dynamics within reconstituted or simplified experimental systems.
Additionally, Aureobasidin A supports computational and structural biology applications, including enzyme–ligand docking, molecular dynamics simulations, and structure–activity relationship (SAR) analysis. These approaches provide complementary insight into IPC synthase inhibition mechanisms and aid in the interpretation of experimental observations at the molecular level.
Due to its defined mechanism, reproducible performance, and high chemical purity, Aureobasidin A is well suited for fundamental research, pathway validation studies, and exploratory screening programs. Supplied as a factory-manufactured, high-purity compound with bulk and wholesale availability, it supports scalable laboratory research projects conducted by academic institutions, biotechnology companies, and pharmaceutical research organizations worldwide.
Research Models
Aureobasidin A is compatible with a wide range of in vitro research models designed to investigate sphingolipid biosynthesis, membrane lipid regulation, and enzyme-specific molecular mechanisms. Its high selectivity for inositol phosphorylceramide (IPC) synthase enables precise interrogation of sphingolipid pathways across multiple experimental platforms while maintaining controlled and reproducible conditions.
One commonly employed research model involves purified or recombinant IPC synthase enzyme systems. In these assays, Aureobasidin A is used to directly assess enzyme activity, inhibition kinetics, and concentration-dependent response profiles. Such enzyme-focused models allow detailed characterization of catalytic parameters and support quantitative comparisons across experimental conditions or compound variants.
Aureobasidin A is also well suited for cell-free lipid metabolism models, including microsomal preparations and reconstituted lipid systems. These models enable examination of sphingolipid flux and ceramide accumulation without interference from complex regulatory networks. By isolating specific biochemical steps, researchers can more clearly define causal relationships within the sphingolipid biosynthesis pathway.
In reconstituted membrane and lipid bilayer models, Aureobasidin A facilitates investigation of how targeted inhibition of IPC synthesis influences membrane lipid composition and physical properties. These systems support studies of lipid packing, microdomain formation, and lipid–protein interactions under defined lipid ratios and environmental conditions.
For omics-integrated research, Aureobasidin A is frequently incorporated into lipidomics-enabled platforms. Controlled pathway modulation allows comprehensive profiling of sphingolipid intermediates using mass spectrometry–based analytical techniques, generating high-resolution datasets for pathway mapping and systems-level interpretation.
Finally, Aureobasidin A is widely applied in computational research models, including enzyme–ligand docking, molecular dynamics simulations, and structure–activity relationship analysis. These in silico models complement experimental data by providing mechanistic insight into binding interactions and inhibitory behavior at the molecular level.
Collectively, these research models establish Aureobasidin A as a versatile and reliable molecular probe for advanced sphingolipid and membrane lipid research conducted exclusively in laboratory-based, in vitro environments.
Experimental Design Considerations
Careful experimental design is essential to fully leverage the specificity and reproducibility of Aureobasidin A in in vitro sphingolipid biosynthesis and membrane lipid regulation studies. Because Aureobasidin A functions as a selective inhibitor of inositol phosphorylceramide (IPC) synthase, experimental systems should be optimized to accurately capture pathway-specific biochemical responses while minimizing confounding variables.
Concentration selection is a primary consideration. Preliminary range-finding experiments are recommended to identify conditions that achieve consistent IPC synthase inhibition without introducing non-specific effects. Preparing stock solutions in appropriate solvents, such as DMSO, and maintaining uniform solvent concentrations across experimental and control groups helps ensure data comparability.
Assay conditions, including buffer composition, temperature, and incubation duration, should be standardized and carefully documented. IPC synthase activity and sphingolipid metabolism are sensitive to physicochemical parameters; therefore, maintaining consistent experimental environments is critical for reproducible results. Inclusion of vehicle-only and baseline controls is strongly recommended to distinguish compound-specific effects from background variation.
For studies involving lipid analysis, integration with validated lipid extraction and quantification workflows is essential. Mass spectrometry–based lipidomics or chromatography-based detection methods enable accurate measurement of ceramide and related sphingolipid intermediates following Aureobasidin A treatment. Proper normalization strategies should be applied to account for variability across samples and experimental runs.
When combining Aureobasidin A with computational or systems-level analyses, experimental outputs should be generated in formats compatible with downstream modeling workflows. Clear documentation of experimental parameters enhances the interpretability of structure–activity relationship studies and pathway simulations.
By adhering to rigorous experimental design principles, researchers can maximize the value of Aureobasidin A as a precise molecular probe for mechanistic investigations conducted exclusively in controlled laboratory research settings.
Laboratory Safety & Handling Guidelines
Aureobasidin A is a research-grade cyclic depsipeptide intended exclusively for in vitro laboratory research. Proper handling, storage, and disposal are essential to ensure both research integrity and laboratory safety.
Handling Precautions
Always use standard laboratory personal protective equipment (PPE), including gloves, lab coat, and safety glasses. Avoid direct contact with skin, eyes, or clothing, and prevent inhalation of powders or aerosols. Work in a well-ventilated area, preferably under a fume hood when preparing concentrated solutions.
Solution Preparation
Dissolve Aureobasidin A in compatible solvents, such as DMSO, to prepare stock solutions. Ensure complete solubilization and avoid repeated freeze–thaw cycles, which can compromise compound integrity. Use precise pipetting and measurement techniques to maintain consistency across experimental replicates.
Storage
Store the compound at −20 °C in a dry, light-protected environment. Protect from moisture and exposure to strong oxidizing agents. Proper storage ensures long-term chemical stability and reproducible experimental performance.
Waste Management
Dispose of all residual compounds, solutions, and contaminated materials according to local chemical waste regulations. Avoid introducing the compound into drains or the environment. Label all containers clearly to prevent accidental misuse.
By following these guidelines, Aureobasidin A can be safely utilized in advanced in vitro sphingolipid biosynthesis and membrane lipid studies, while preserving its high-purity and functional integrity for reproducible research outcomes.
Integration with Multi-Omic & Computational Studies
Aureobasidin A is ideally suited for integration with multi-omic platforms and computational modeling approaches, enhancing the depth and precision of sphingolipid research. Its selective inhibition of inositol phosphorylceramide (IPC) synthase allows controlled perturbation of sphingolipid metabolism, which can be directly correlated with lipidomic, proteomic, and systems-level molecular data.
In lipidomics workflows, Aureobasidin A facilitates detailed profiling of IPC depletion, ceramide accumulation, and downstream sphingolipid intermediates using mass spectrometry or chromatography-based detection methods. This enables researchers to map pathway flux and quantify specific lipid species with high resolution. Complementary proteomics studies can assess lipid-regulated protein interactions, membrane-associated signaling changes, and structural protein dynamics, providing mechanistic insight into lipid-dependent molecular processes.
Aureobasidin A is also compatible with computational approaches, including enzyme–ligand docking, molecular dynamics simulations, and structure–activity relationship (SAR) analysis. These in silico techniques complement experimental data by modeling IPC synthase binding interactions, predicting inhibitory potency, and guiding hypothesis-driven research.
By combining biochemical, lipidomic, proteomic, and computational data, researchers can construct an integrated understanding of sphingolipid regulation, membrane organization, and pathway-specific responses. This systems-level approach maximizes mechanistic insight and supports reproducible, high-confidence conclusions in in vitro mechanistic studies.
Keywords
Aureobasidin A, IPC synthase inhibitor, sphingolipid biosynthesis, in vitro lipid metabolism, membrane lipid regulation, cyclic depsipeptide inhibitor, IPC pathway research, lipidomics reagent, high-purity Aureobasidin A, factory manufactured research compound, China B2B wholesale
Shipping Guarantee
Aureobasidin A is shipped in secure, laboratory-grade packaging to protect product integrity during transit. Temperature-sensitive logistics are applied when necessary, and all shipments include tracking and confirmation of delivery. We ensure fast, reliable, and traceable delivery worldwide, minimizing delays for research projects.
Trade Assurance
As a factory-direct manufacturer, we provide high-purity, batch-verified Aureobasidin A with accompanying Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS). Bulk and wholesale supply options are available to support long-term research needs. Our quality-controlled production ensures reproducibility and consistency across shipments.
Payment Support
We accept multiple secure payment methods to facilitate global B2B transactions. Options include credit cards (Visa, MasterCard, AmEx), telegraphic transfers (TT/SWIFT), and encrypted cryptocurrency payments, as well as Alipay and WeChat Pay. All payments are processed through secure channels to ensure confidentiality and reliability. Bulk and wholesale orders are supported with flexible terms to accommodate research institutions and companies.
Disclaimer
Aureobasidin A is intended exclusively for laboratory research use. It is not for human or veterinary use, and must not be applied in clinical, diagnostic, or therapeutic settings. Researchers must comply with all local regulations and laboratory safety protocols when handling this compound.
References
Aureobasidin A inhibits yeast IPC synthase — Demonstrates that Aureobasidin A targets inositol phosphorylceramide synthase (Aur1p) and alters sphingolipid levels, including ceramide accumulation. PubMed
Fluorometric assay characterization of IPC synthase inhibition — Provides experimental evidence of IPC synthase inhibition by Aureobasidin A with kinetic analysis in vitro. PubMed
IPC synthase inhibition in Candida and Aspergillus models — Shows that IPC synthase activity across fungal species is sensitive to Aureobasidin A-mediated inhibition in membrane preparations. PubMed
Cryo‑EM elucidation of Aureobasidin A bound to IPC synthase complex — Recent structural study detailing how Aureobasidin A occupies the enzyme active site and blocks substrate entry, enhancing mechanistic insight. Nature
Functional effects of Aureobasidin A on Toxoplasma sphingolipid synthesis — Open‑access PMC article confirming Aureobasidin A’s interaction with IPC synthase pathways in protozoan models, supporting its biochemical mechanism. pmc.ncbi.nlm.nih.g
Additional information
| Weight | 1 kg |
|---|---|
| Dimensions | 26 × 23 × 26 cm |
2 reviews for Aureobasidin A – 99%High-purity peptides
What is Aureobasidin A used for in research?
Aureobasidin A is a selective inhibitor of inositol phosphorylceramide (IPC) synthase, used in in vitro studies of sphingolipid biosynthesis, membrane lipid regulation, and enzyme-specific pathway analysis.
Does Aureobasidin A specifically inhibit IPC synthase?
Yes. Its cyclic depsipeptide structure allows highly selective binding, enabling precise pathway-specific inhibition without broadly affecting unrelated enzymes.
Is Aureobasidin A suitable for lipidomics studies?
Absolutely. It is commonly used in mass spectrometry-based lipidomics workflows to track ceramide accumulation and IPC depletion in controlled experimental systems.
What purity level is provided?
Aureobasidin A is supplied at ≥98–99% purity (HPLC verified), ensuring reproducible biochemical and mechanistic results.
How should Aureobasidin A be stored?
Store at −20 °C, protected from light and moisture, in a dry, stable environment to maintain chemical integrity.
What solvents are recommended?
DMSO is the recommended solvent for stock solutions. Limited solubility in aqueous buffers is expected; proper dilution and mixing are advised.
Can Aureobasidin A be used in enzyme inhibition assays?
Yes. It is ideal for IPC synthase inhibition assays, including kinetics, dose-response studies, and structure-activity relationship evaluations.
Is it compatible with membrane reconstitution studies?
Yes. Researchers use Aureobasidin A in reconstituted lipid bilayers to investigate sphingolipid-dependent membrane organization and microdomain formation.
Can it be combined with computational studies?
Yes. Its defined cyclic depsipeptide structure allows molecular docking, dynamics simulations, and enzyme-ligand modeling.
Does it support bulk and wholesale supply?
Yes. Factory-direct production supports high-purity bulk supply, low-cost wholesale, and global B2B export.
Are COA and MSDS provided?
Yes. Each batch includes Certificate of Analysis (COA) and Material Safety Data Sheet (MSDS) for laboratory compliance and quality assurance.
Is it suitable for long-term research projects?
Yes. High purity, consistent batch quality, and proper storage enable long-term reproducible use in multiple experimental setups.
How does Aureobasidin A affect ceramide accumulation?
By inhibiting IPC synthase, Aureobasidin A causes accumulation of upstream ceramide intermediates, which can be measured in lipid profiling studies.
Which in vitro models are compatible?
Purified IPC synthase, cell-free lipid metabolism systems, reconstituted membrane models, and multi-omic compatible workflows are all appropriate.









marilynrnc –
The ordering process was smooth from inquiry to delivery.
Naccion –
The integrity of the service attitude moved me.