No products in the cart.
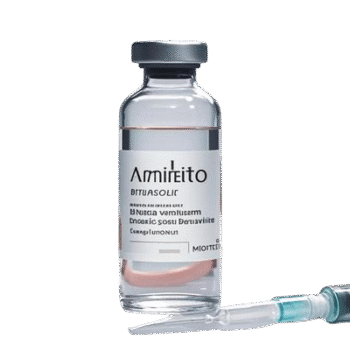
Vyvgart Efgartigimod Alfa Injection 400?mg/20?ml – (For Research Use Only)
$10.00
Vyvgart Efgartigimod Alfa Injection is a human IgG1 Fc fragment that blocks the neonatal Fc receptor (FcRn) to reduce circulating IgG levels. Supplied as 400?mg in 20?ml sterile solution. Manufactured by Vetter Pharma?Fertigung GmbH under Chinese approval SJ20230008. Ideal for laboratory research on IgG-mediated autoimmune mechanisms, immunology modeling, and FcRn pharmacodynamics. Wholesale and retail options available. For research use only.?For wholesale prices, other specifications and uses, please consult our staff?
Description
Vyvgart Efgartigimod Alfa is an engineered Fc fragment that binds with high affinity to FcRn, preventing IgG recycling and thereby accelerating IgG degradation. This results in a marked and sustained reduction in total IgG, including pathogenic autoantibodies. Clinically approved for generalized myasthenia gravis and chronic inflammatory demyelinating polyneuropathy in China, this formulation serves as a powerful tool in research settings for modeling IgG?mediated autoimmune conditions, exploring FcRn pharmacology, and developing antibody clearance interventions. Each vial contains 400?mg efgartigimod alfa in a buffered injectable solution. Strictly for laboratory research use only.
Product Specifications
| Parameter | Detail |
|---|---|
| Product Name | Vyvgart Efgartigimod Alfa Injection |
| Strength & Volume | 400?mg per 20?ml vial |
| Dosage Form | Sterile intravenous or subcutaneous injectable Fc fragment |
| Packaging Unit | 1 vial per box |
| Manufacturer | Vetter Pharma?Fertigung GmbH & Co. KG (Germany) |
| Approval Number | ???? SJ20230008 |
| Drug Standard Code | 86984830000013 |
| Barcode | Not yet assigned |
| CAS Number | 1821402?21?4 |
| Molecular Weight | ~54?kDa protein fragment |
Mechanism of Action & Research Applications
Vyvgart functions as a neonatal Fc receptor blocker: it competes with endogenous IgG for FcRn binding, preventing IgG recycling and promoting lysosomal degradation. This targeted reduction in IgG—including autoantibodies—makes it an excellent model for studying FcRn biology, autoantibody-associated conditions such as myasthenia gravis or CIDP, and therapeutic antibody removal kinetics. It has broad utility in immunology, neurology, and immunotherapy research.
Side Effects (For Reference Only in Research Models)
In clinical and analogous model observations, potential effects include mild respiratory infections, headaches, urinary tract infections, paresthesia (tingling/numbness), and occasional infusion site discomfort. These are typically transient. Monitor inflammatory markers and immune cell profiles during experimental protocols.
Disclaimer
Vyvgart Efgartigimod Alfa Injection is strictly intended for laboratory research use only. Not for human or veterinary therapeutic or diagnostic application.
Additional information
| Weight | 0.6 kg |
|---|---|
| Dimensions | 23 × 36 × 23 cm |







Reviews
There are no reviews yet.